Just putting this here in case anyone is still subscribed. Due to issues with hosting requirements, the Variety of Life website has changed its address. It can now be found at https://varietyoflife.net/
- Home
- Angry by Choice
- Catalogue of Organisms
- Chinleana
- Doc Madhattan
- Games with Words
- Genomics, Medicine, and Pseudoscience
- History of Geology
- Moss Plants and More
- Pleiotropy
- Plektix
- RRResearch
- Skeptic Wonder
- The Culture of Chemistry
- The Curious Wavefunction
- The Phytophactor
- The View from a Microbiologist
- Variety of Life
Field of Science
-
-
Change of address10 months ago in Variety of Life
-
Change of address10 months ago in Catalogue of Organisms
-
-
Earth Day: Pogo and our responsibility1 year ago in Doc Madhattan
-
What I Read 20241 year ago in Angry by Choice
-
I've moved to Substack. Come join me there.1 year ago in Genomics, Medicine, and Pseudoscience
-
-
-
-
Histological Evidence of Trauma in Dicynodont Tusks7 years ago in Chinleana
-
Posted: July 21, 2018 at 03:03PM7 years ago in Field Notes
-
Why doesn't all the GTA get taken up?7 years ago in RRResearch
-
-
Harnessing innate immunity to cure HIV9 years ago in Rule of 6ix
-
-
-
-
-
-
post doc job opportunity on ribosome biochemistry!11 years ago in Protein Evolution and Other Musings
-
Blogging Microbes- Communicating Microbiology to Netizens11 years ago in Memoirs of a Defective Brain
-
Re-Blog: June Was 6th Warmest Globally11 years ago in The View from a Microbiologist
-
-
-
The Lure of the Obscure? Guest Post by Frank Stahl13 years ago in Sex, Genes & Evolution
-
-
Lab Rat Moving House14 years ago in Life of a Lab Rat
-
Goodbye FoS, thanks for all the laughs14 years ago in Disease Prone
-
-
Slideshow of NASA's Stardust-NExT Mission Comet Tempel 1 Flyby15 years ago in The Large Picture Blog
-
in The Biology Files
Catalogue of Organisms
An inordinate fondness for systematics
The Site is Dead, Long Live the Site
I'm moving house.
Over the past few years, Blogger has become somewhat less user-friendly behind the scenes. Nothing major, and certainly nothing I'm going to bore you with here, but enough that I've finally decided to take the step of breaking out and moving to my own site: varietyoflife.net. This site will incorporate material from both Catalogue of Organisms and Variety of Life, progressively merging them into a single guide to global biodiversity. Posts from both blogs have already been migrated over, though now I have the long task of editing and updating them to match the new format. In the meantime, check out the page on Prostigmata to get an idea of what I'm planning to do. New content will also be appearing regularly.
Thank you for reading Catalogue of Organisms, and I hope to see you in the new digs!
Over the past few years, Blogger has become somewhat less user-friendly behind the scenes. Nothing major, and certainly nothing I'm going to bore you with here, but enough that I've finally decided to take the step of breaking out and moving to my own site: varietyoflife.net. This site will incorporate material from both Catalogue of Organisms and Variety of Life, progressively merging them into a single guide to global biodiversity. Posts from both blogs have already been migrated over, though now I have the long task of editing and updating them to match the new format. In the meantime, check out the page on Prostigmata to get an idea of what I'm planning to do. New content will also be appearing regularly.
Thank you for reading Catalogue of Organisms, and I hope to see you in the new digs!
Bouncing Snail-y Clams
For the most part, bivalves are a fairly conservative bunch. They seem to have worked out what they are good at early on in their history and most of them stick to it. There are, however, notable exceptions and perhaps few groups of bivalves are as exceptional as the Galeommatidae.
Waldo paucitentaculatus, from Valentich-Scott et al. (2013).
Galeommatids are small bivalves, less than a centimetre in length, with more or less thin shells. The hinge teeth are generally weak or absent. The valves of the shell are more or less gaping and in life are at least partially covered by the large, reflected mantle. The outer surface of the mantle is warty and bears several slender tentacles, the exact arrangement of tentacles varying by species. The foot is large and extends well outwards from the central body of the animal. In the most extreme cases, you might be forgiven for thinking you were looking at some sort of snail rather than a clam.
Many galeommatids have been found living as symbionts with other invertebrates such as in the burrows of annelids and crustaceans, or crawling on the surface of echinoderms. So far as is known, these relationships are commensal only, the clams using their hosts as a source of shelter and possibly excess food scraps, but species may be very exclusive in their choice of hosts. For instance, Mikkelsen & Bieler (1989) found the species Divariscintilla yoyo and D. troglodytes only in burrows of the mantis shrimp Lysiosquilla scabricauda, never in burrows of other potential hosts in the same area. It seems likely that this commensalism has allowed galeommatids to diversify in soft-bottom habitats, their larger hosts being able to dig into sediments in which the smaller clams would be quickly smothered (Valentich-Scott et al. 2013).
Unidentified galeommatid, copyright Ria Tan.
Many galeommatids possess a distinctive 'hanging-foot' morphology with the foot divided into two sections, a muscular anterior portion adapted for snail-like crawling, and an elastic posterior section (Bieler & Mikkelsen 1992). The primary byssus gland is located in the anterior section and is connected by a ciliated ventral groove to a terminal adhesive gland in the posterior section. Mikkelsen & Bieler (1989) found that Divariscintilla individuals kept in an aquarium spent most of their time hanging suspended via the posterior part of the foot. Threads produced by the byssus gland were transferred to the terminal adhesor and used to attach to a surface such as the glass of the aquarium (presumably, the clams would normally hang in this manner on the interior wall of the host burrow). When disturbed, hanging clams would rapidly bounce themselves up and down from their attachment point (hence one species being dubbed 'Divariscintilla yoyo'). If the clams wished to change their location, they would crawl on the muscular section of the foot, breaking the byssus threads behind them. The elastic part of the foot was not functional in crawling.
The majority of galeommatid clams are hermaphrodites, either protandrous (beginning life as males before maturing into females) or simultaneous. Eggs are not released into the water column but brooded within the ctenidia until larvae are released at a relatively advanced stage of development (whether the parent is able to feed while its gills are so occupied, I don't know). In a number of species, dwarf males are also present that do not live independently but reside within the mantle cavity of a female (I have seen these males referred to as 'parasitic' but I do not know if they are directly so). In this position, they are able to fertilise the female directly. Such behaviour may be seen as a further adaptation to the clam's commensal lifestyle, contained within the burrow of its host and potentially secluded from more conventional mates. Hidden away in the darkness, they make matryoshkas of themselves.
REFERENCES
Bieler, R., & P. M. Mikkelsen. 1992. Preliminary phylogenetic analysis of the bivalve family Galeommatidae. American Malacological Bulletin 9 (2): 157–164.
Mikkelsen, P. M., & R. Bieler. 1989. Biology and comparative anatomy of Divariscintilla yoyo and D. troglodytes, two new species of Galeommatidae (Bivalvia) from stomatopod burrows in eastern Florida. Malacologia 31 (1): 175–195.
Valentich-Scott, P., D. Ó. Foighil & J. Li. 2013. Where's Waldo? A new commensal species, Waldo arthuri (Mollusca, Bivalvia, Galeommatidae) from the northeastern Pacific Ocean. ZooKeys 316: 67–80.
Galeommatids are small bivalves, less than a centimetre in length, with more or less thin shells. The hinge teeth are generally weak or absent. The valves of the shell are more or less gaping and in life are at least partially covered by the large, reflected mantle. The outer surface of the mantle is warty and bears several slender tentacles, the exact arrangement of tentacles varying by species. The foot is large and extends well outwards from the central body of the animal. In the most extreme cases, you might be forgiven for thinking you were looking at some sort of snail rather than a clam.
Many galeommatids have been found living as symbionts with other invertebrates such as in the burrows of annelids and crustaceans, or crawling on the surface of echinoderms. So far as is known, these relationships are commensal only, the clams using their hosts as a source of shelter and possibly excess food scraps, but species may be very exclusive in their choice of hosts. For instance, Mikkelsen & Bieler (1989) found the species Divariscintilla yoyo and D. troglodytes only in burrows of the mantis shrimp Lysiosquilla scabricauda, never in burrows of other potential hosts in the same area. It seems likely that this commensalism has allowed galeommatids to diversify in soft-bottom habitats, their larger hosts being able to dig into sediments in which the smaller clams would be quickly smothered (Valentich-Scott et al. 2013).
Many galeommatids possess a distinctive 'hanging-foot' morphology with the foot divided into two sections, a muscular anterior portion adapted for snail-like crawling, and an elastic posterior section (Bieler & Mikkelsen 1992). The primary byssus gland is located in the anterior section and is connected by a ciliated ventral groove to a terminal adhesive gland in the posterior section. Mikkelsen & Bieler (1989) found that Divariscintilla individuals kept in an aquarium spent most of their time hanging suspended via the posterior part of the foot. Threads produced by the byssus gland were transferred to the terminal adhesor and used to attach to a surface such as the glass of the aquarium (presumably, the clams would normally hang in this manner on the interior wall of the host burrow). When disturbed, hanging clams would rapidly bounce themselves up and down from their attachment point (hence one species being dubbed 'Divariscintilla yoyo'). If the clams wished to change their location, they would crawl on the muscular section of the foot, breaking the byssus threads behind them. The elastic part of the foot was not functional in crawling.
The majority of galeommatid clams are hermaphrodites, either protandrous (beginning life as males before maturing into females) or simultaneous. Eggs are not released into the water column but brooded within the ctenidia until larvae are released at a relatively advanced stage of development (whether the parent is able to feed while its gills are so occupied, I don't know). In a number of species, dwarf males are also present that do not live independently but reside within the mantle cavity of a female (I have seen these males referred to as 'parasitic' but I do not know if they are directly so). In this position, they are able to fertilise the female directly. Such behaviour may be seen as a further adaptation to the clam's commensal lifestyle, contained within the burrow of its host and potentially secluded from more conventional mates. Hidden away in the darkness, they make matryoshkas of themselves.
REFERENCES
Bieler, R., & P. M. Mikkelsen. 1992. Preliminary phylogenetic analysis of the bivalve family Galeommatidae. American Malacological Bulletin 9 (2): 157–164.
Mikkelsen, P. M., & R. Bieler. 1989. Biology and comparative anatomy of Divariscintilla yoyo and D. troglodytes, two new species of Galeommatidae (Bivalvia) from stomatopod burrows in eastern Florida. Malacologia 31 (1): 175–195.
Valentich-Scott, P., D. Ó. Foighil & J. Li. 2013. Where's Waldo? A new commensal species, Waldo arthuri (Mollusca, Bivalvia, Galeommatidae) from the northeastern Pacific Ocean. ZooKeys 316: 67–80.
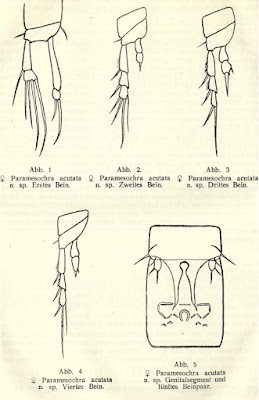
Paramesochra acutata
Copepod taxonomy, it seems, is largely about counting setae. In his review of relationships within the interstitial harpacticoid family Paramesochridae, Huys (1987) recognised four species groups within the genus Paramesochra (which previously got a look-in at this site here). One of these groups, labelled the P. acutata-group, was characterised by reductions in numbers of setae, having lost the inner setae on the first segments of the endopods on the third and fourth legs.
Paramesochra taeana, a close relative of P. acutata, from Back & Lee (2010).
The group takes its name from the species Paramesochra acutata, described by Klie in 1935 from samples taken from coastal groundwater near the town of Schilksee on the northeastern coast of Germany, in the state of Schleswig-Holstein. Other notable features of P. acutata include the presence of four setae on the antennary exopod, well-developed narrow, triangular endopodal lobes on the modified fifth legs of the females, and conical caudal rami produced into spinose processes (Back & Lee 2013). I haven't been able to find whether P. acutata has been collected much beyond its initial locality but other members of its species group have been found around the world. One of these, P. hawaiensis, from (nach) Hawaii, is similar enough that it was until recently treated as a subspecies of P. acutata.
Appendages of female Paramesochra acutata, from Klie (1935).
So what, if anything, does all this mean? That, I'm afraid, is getting a bit beyond me. The fifth legs are used in spermatophore transfer and differences between species might presumably function in recognising suitable mates. Regarding the details of setation and ramus appearance, one wonders if there could be any relation to preferred micro-habitat. Are harpacticoids with fewer setae and more robust rami adapted for crawling among coarser sand grains? Honestly, I have no idea. Anyone care to find out?
REFERENCES
Back, J., & W. Lee. 2010. A new species of the genus Paramesochra (Copepoda: Harpacticoida) from Korean waters. Proceedings of the Biological Society of Washington 123 (1): 47–61.
Back, J., & W. Lee. 2013. Three new species of the genus Paramesochra T. Scott, 1892 (Copepoda: Harpacticoida: Paramesochridae) from Yellow Sea, Korea with a redescription of Paramesochra similis Kunz, 1936. Journal of Natural History 47 (5–12): 769–803.
Huys, R. 1987. Paramesochra T. Scott, 1892 (Copepoda, Harpacticoida): a revised key, including a new species from the SW Dutch coast and some remarks on the phylogeny of the Paramesochridae. Hydrobiologia 144: 193–210.
Klie, W. 1935. Die Harpacticoiden des Küstengrundwassers bei Schilksee (Kieler Förde). Schriften des Naturwissenschaftlichen Vereins für Schleswig-Holstein 20 (2): 409–421.
The group takes its name from the species Paramesochra acutata, described by Klie in 1935 from samples taken from coastal groundwater near the town of Schilksee on the northeastern coast of Germany, in the state of Schleswig-Holstein. Other notable features of P. acutata include the presence of four setae on the antennary exopod, well-developed narrow, triangular endopodal lobes on the modified fifth legs of the females, and conical caudal rami produced into spinose processes (Back & Lee 2013). I haven't been able to find whether P. acutata has been collected much beyond its initial locality but other members of its species group have been found around the world. One of these, P. hawaiensis, from (nach) Hawaii, is similar enough that it was until recently treated as a subspecies of P. acutata.
So what, if anything, does all this mean? That, I'm afraid, is getting a bit beyond me. The fifth legs are used in spermatophore transfer and differences between species might presumably function in recognising suitable mates. Regarding the details of setation and ramus appearance, one wonders if there could be any relation to preferred micro-habitat. Are harpacticoids with fewer setae and more robust rami adapted for crawling among coarser sand grains? Honestly, I have no idea. Anyone care to find out?
REFERENCES
Back, J., & W. Lee. 2010. A new species of the genus Paramesochra (Copepoda: Harpacticoida) from Korean waters. Proceedings of the Biological Society of Washington 123 (1): 47–61.
Back, J., & W. Lee. 2013. Three new species of the genus Paramesochra T. Scott, 1892 (Copepoda: Harpacticoida: Paramesochridae) from Yellow Sea, Korea with a redescription of Paramesochra similis Kunz, 1936. Journal of Natural History 47 (5–12): 769–803.
Huys, R. 1987. Paramesochra T. Scott, 1892 (Copepoda, Harpacticoida): a revised key, including a new species from the SW Dutch coast and some remarks on the phylogeny of the Paramesochridae. Hydrobiologia 144: 193–210.
Klie, W. 1935. Die Harpacticoiden des Küstengrundwassers bei Schilksee (Kieler Förde). Schriften des Naturwissenschaftlichen Vereins für Schleswig-Holstein 20 (2): 409–421.
Melanterius Weevils
Here in the Antipodes, we have a long history of environmental upheaval from exotic taxa unwisely released. As a result, one can't help but feel an odd twinge of perverse patriotism when hearing of the inverse, some native of the Antipodes causing grief elsewhere. In South Africa, Australian acacias have become something of an issue, inciting a search for potential control agents. Among the candidates selected are weevils of the genus Melanterius.
Melanterius servulus, copyright Sally Adam.
Melanterius is a diverse genus of small black or brown weevils (ranging from about three to seven millimetres in length) that feed as both adults and larvae on the developing seeds of acacias. About eighty species have been recognised in the genus to date and possibly many more remain to be described. In general, Melanterius weevils are heavily punctate, usually without prominent hairs but with a covering of scales. The rostrum is reasonably long, reaching more or less back to the mesosternum at rest but not sitting in a distinct ventral groove, and may be variably curved (going by figures in Zimmerman 1992).
Melanterius semiporcatus, copyright Victor W. Fazio III.
As with other weevils, the prominent rostrum is used by females to chew into an appropriate spot on the host plant, in this case chewing holes into the developing acacia seed pods, into which eggs are laid. Melanterius species go through one generation per year. Larvae burrow into and feed on the developing seeds before emerging and dropping to the ground to pupate in the soil. Mature adults emerge well before the host acacias begin to set seeds, usually having to wait about six months (Auld 1989). They usually spend the intervening period largely inactive, sheltering in concealed places close to the host plant and occasionally emerging to briefly feed on developing buds.
Under peak conditions, Melanterius infestations may cause a complete failure of seed production. No wonder, then, that they have been considered a worthwhile instrument of biological control.
REFERENCES
Auld, T. D. 1989. Larval survival in the soil and adult emergence in Melanterius Erichson and Plaesiorhinus Blackburn (Coleoptera: Curculionidae) following seed feeding on Acacia and Bossiaea (Fabaceae). Journal of the Australian Entomological Society 28: 235–238.
Zimmerman, E. C. 1992. Australian Weevils (Coleoptera: Curculionoidea) vol. 6. Colour plates 305–632. CSIRO Australia.
Melanterius is a diverse genus of small black or brown weevils (ranging from about three to seven millimetres in length) that feed as both adults and larvae on the developing seeds of acacias. About eighty species have been recognised in the genus to date and possibly many more remain to be described. In general, Melanterius weevils are heavily punctate, usually without prominent hairs but with a covering of scales. The rostrum is reasonably long, reaching more or less back to the mesosternum at rest but not sitting in a distinct ventral groove, and may be variably curved (going by figures in Zimmerman 1992).
As with other weevils, the prominent rostrum is used by females to chew into an appropriate spot on the host plant, in this case chewing holes into the developing acacia seed pods, into which eggs are laid. Melanterius species go through one generation per year. Larvae burrow into and feed on the developing seeds before emerging and dropping to the ground to pupate in the soil. Mature adults emerge well before the host acacias begin to set seeds, usually having to wait about six months (Auld 1989). They usually spend the intervening period largely inactive, sheltering in concealed places close to the host plant and occasionally emerging to briefly feed on developing buds.
Under peak conditions, Melanterius infestations may cause a complete failure of seed production. No wonder, then, that they have been considered a worthwhile instrument of biological control.
REFERENCES
Auld, T. D. 1989. Larval survival in the soil and adult emergence in Melanterius Erichson and Plaesiorhinus Blackburn (Coleoptera: Curculionidae) following seed feeding on Acacia and Bossiaea (Fabaceae). Journal of the Australian Entomological Society 28: 235–238.
Zimmerman, E. C. 1992. Australian Weevils (Coleoptera: Curculionoidea) vol. 6. Colour plates 305–632. CSIRO Australia.
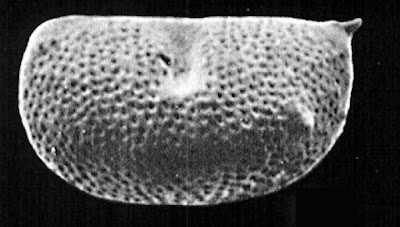
Kirkby's Small Ostracods (or Small Kirkby's Ostracods)
I do not envy those who find themselves working with ostracods. These minute crustaceans, typically less than a millimetre in length, seem altogether too fiddly to handle. Nevertheless, the long history of ostracods, together with their diversity and the high fossilisation potential of their calcified carapace valves, have made them a common focus for studying biostratigraphy and historical environments. The classification of modern ostracods is commonly informed by features of the legs and other appendages but such characters are not commonly preserved in fossil representatives. As a result, there are many groups of ostracods known from the Palaeozoic whose relationships remain uncertain.
Left valve of Kirkbyella delicata, from Hoare & Merrill (2004).
One such group is classified by Liebau (2005) as the superfamily Kirkbyelloidea. Members of this group are small ostracods with reticulate valves. The dorsal and ventral margins of the valves tend to be more or less straight. They are commonly impressed with a single dorsal sulcus, extending downwards from the dorsal margin about halfway along the valve's length. Below this sulcus is a protruding horizontal lobe ending in members of the family Kirkbyellidae in a small spine. Evidence of sexual dimorphism, a not-uncommon feature of Palaeozoic ostracods, is not known from kirkbyelloids.
Definite kirkbyelloids are known from the Devonian to the Permian. If the earlier family Ordovizonidae is included, their record extends all the way back to the Ordovician. As noted above, it is unclear where kirkbyelloids sit in the ostracod family tree. Becker (1994) suggested a relationship via Ordovizona to the Ordovician Monotiopleuridae which resemble kirkbyelloids in the outline of the carapace valves and features of the adductor muscle scars. Though long-lived, kirkbyelloids don't seem to have ever been massively diverse, and they can probably be counted among the many lineages of organisms that never made it past the end of the Palaeozoic.
REFERENCES
Becker, G. 1994. A remarkable Ordovician ostracod fauna from Orphan Knoll, Labrador Sea. Scripta Geologica 107: 1–25.
Hoare, R. D., & G. K. Merrill. 2004. A Pennsylvanian (Morrowan) ostracode fauna from Texas. Journal of Paleontology 78 (1): 185–204.
Liebau, A. 2005. A revised classification of the higher taxa of the Ostracoda (Crustacea). Hydrobiologia 538: 115–137.
One such group is classified by Liebau (2005) as the superfamily Kirkbyelloidea. Members of this group are small ostracods with reticulate valves. The dorsal and ventral margins of the valves tend to be more or less straight. They are commonly impressed with a single dorsal sulcus, extending downwards from the dorsal margin about halfway along the valve's length. Below this sulcus is a protruding horizontal lobe ending in members of the family Kirkbyellidae in a small spine. Evidence of sexual dimorphism, a not-uncommon feature of Palaeozoic ostracods, is not known from kirkbyelloids.
Definite kirkbyelloids are known from the Devonian to the Permian. If the earlier family Ordovizonidae is included, their record extends all the way back to the Ordovician. As noted above, it is unclear where kirkbyelloids sit in the ostracod family tree. Becker (1994) suggested a relationship via Ordovizona to the Ordovician Monotiopleuridae which resemble kirkbyelloids in the outline of the carapace valves and features of the adductor muscle scars. Though long-lived, kirkbyelloids don't seem to have ever been massively diverse, and they can probably be counted among the many lineages of organisms that never made it past the end of the Palaeozoic.
REFERENCES
Becker, G. 1994. A remarkable Ordovician ostracod fauna from Orphan Knoll, Labrador Sea. Scripta Geologica 107: 1–25.
Hoare, R. D., & G. K. Merrill. 2004. A Pennsylvanian (Morrowan) ostracode fauna from Texas. Journal of Paleontology 78 (1): 185–204.
Liebau, A. 2005. A revised classification of the higher taxa of the Ostracoda (Crustacea). Hydrobiologia 538: 115–137.
Conformed Flycatchers
A quote I have often had cause to refer to—I believe it originally came from Toby White of Palaeos.com—is that "organisms are under no obligation to speciate with regard to the convenience of taxonomists". For birdwatchers in North America, perhaps no group more embodies this principle than the flycatchers of the genus Empidonax. These small members of the hyperdiverse New World family Tyrannidae comprise fifteen recognised species that have become notorious for the difficulty in telling them apart.
Immature alder flycatcher Empidonax alnorum, copyright Cephas.
The species of Empidonax are uniformly olive brown above, lighter below, with pale rings around the eyes and bands on the wings. They are inhabitants of woodlands (more on that in a moment) and watch for flying insects from a perch, making short flights to capture prey. Though individual species are generally similar in their feeding habits, they are often specifically distinct in their preferred habitats. A molecular (mtDNA) analysis of Empidonax species by Johnson & Cicero (2002) identified four likely clades within the genus with members of a clade each differing in their specific breeding range. Species found in the US and Canada often migrate long distances and closely related species may be found close together outside their breeding ranges (references to ranges below refer to breeding ranges). Species found in Mexico and Central America are more likely to migrate only short distances or be resident year-round.
Acadian flycatcher Empidonax virescens, copyright Aitor.
The Acadian flycatcher E. virescens seems to be relatively isolated from other members of the genus. This species is found in shady forests near water in the eastern US and Canada. Its nest is a cup made from plant fibres suspended in a horizontal branch fork, and it lays lightly speckled eggs.
The yellow-bellied flycatcher E. flaviventris, yellowish flycatcher E. flavescens, Cordilleran flycatcher E. occidentalis and Pacific slope flycatcher E. difficilis form a clade of species that tend to have more yellowish underparts than other members of the genus. Their nests are mossy cups constructed on a protected ledge or crevice. Members of this clade tend to be found in relatively damp forest areas, such as boggy areas of boreal forests in the case of E. flaviventris, or shady canyons in the case of E. occidentalis or E. difficilis. A notable exception is the Channel Islands population of E. difficilis which is found in more open woodlands than its mainland counterparts. Empidonax occidentalis and E. difficilis are found in the western United States with E. difficilis occupying coastal regions and E. occidentalis found further inland. Until fairly recently, the two were confused as a single species; they are almost indistinguishable morphologically but can be separated by their calls.
Least flycatcher Empidonax minimus, copyright Mdf.
The white-throated flycatcher E. albigularis, alder flycatcher E. alnorum and willow flycatcher E. traillii form a clade of species nesting in damp thickets. Again, it was only fairly recently that the more northerly E. alnorum was distinguished from the more southerly E. traillii.
Finally, the remaining species form a clade whose members lay eggs without speckled markings. They are often relatively dark compared to other Empidonax; the black-capped flycatcher E. atriceps of Costa Rica and Panama stands out for the sooty-black coloration of the head. They often inhabit relatively open forest, often at higher altitudes.
Johnson & Cicero (2002) suggested that the largely allopatric (non-overlapping) breeding ranges of species within clades of Empidonax reflected speciation as a result of isolation in glacial refuges during the ice ages. As the ice retreated, the now-distinct species expanded their ranges but excluded each other where they met. Differences in mating calls between related species dissuaded interbreeding. Physical appearance, meanwhile, remained frustratingly monotonous.
REFERENCE
Johnson, N. K., & C. Cicero. 2002. The role of ecologic diversification in sibling speciation of Empidonax flycatchers (Tyrannidae): multigene evidence from mtDNA. Molecular Ecology 11: 2065–2081.
The species of Empidonax are uniformly olive brown above, lighter below, with pale rings around the eyes and bands on the wings. They are inhabitants of woodlands (more on that in a moment) and watch for flying insects from a perch, making short flights to capture prey. Though individual species are generally similar in their feeding habits, they are often specifically distinct in their preferred habitats. A molecular (mtDNA) analysis of Empidonax species by Johnson & Cicero (2002) identified four likely clades within the genus with members of a clade each differing in their specific breeding range. Species found in the US and Canada often migrate long distances and closely related species may be found close together outside their breeding ranges (references to ranges below refer to breeding ranges). Species found in Mexico and Central America are more likely to migrate only short distances or be resident year-round.
The Acadian flycatcher E. virescens seems to be relatively isolated from other members of the genus. This species is found in shady forests near water in the eastern US and Canada. Its nest is a cup made from plant fibres suspended in a horizontal branch fork, and it lays lightly speckled eggs.
The yellow-bellied flycatcher E. flaviventris, yellowish flycatcher E. flavescens, Cordilleran flycatcher E. occidentalis and Pacific slope flycatcher E. difficilis form a clade of species that tend to have more yellowish underparts than other members of the genus. Their nests are mossy cups constructed on a protected ledge or crevice. Members of this clade tend to be found in relatively damp forest areas, such as boggy areas of boreal forests in the case of E. flaviventris, or shady canyons in the case of E. occidentalis or E. difficilis. A notable exception is the Channel Islands population of E. difficilis which is found in more open woodlands than its mainland counterparts. Empidonax occidentalis and E. difficilis are found in the western United States with E. difficilis occupying coastal regions and E. occidentalis found further inland. Until fairly recently, the two were confused as a single species; they are almost indistinguishable morphologically but can be separated by their calls.
The white-throated flycatcher E. albigularis, alder flycatcher E. alnorum and willow flycatcher E. traillii form a clade of species nesting in damp thickets. Again, it was only fairly recently that the more northerly E. alnorum was distinguished from the more southerly E. traillii.
Finally, the remaining species form a clade whose members lay eggs without speckled markings. They are often relatively dark compared to other Empidonax; the black-capped flycatcher E. atriceps of Costa Rica and Panama stands out for the sooty-black coloration of the head. They often inhabit relatively open forest, often at higher altitudes.
Johnson & Cicero (2002) suggested that the largely allopatric (non-overlapping) breeding ranges of species within clades of Empidonax reflected speciation as a result of isolation in glacial refuges during the ice ages. As the ice retreated, the now-distinct species expanded their ranges but excluded each other where they met. Differences in mating calls between related species dissuaded interbreeding. Physical appearance, meanwhile, remained frustratingly monotonous.
REFERENCE
Johnson, N. K., & C. Cicero. 2002. The role of ecologic diversification in sibling speciation of Empidonax flycatchers (Tyrannidae): multigene evidence from mtDNA. Molecular Ecology 11: 2065–2081.
Subscribe to:
Posts (Atom)