Yours truly has just spent an evening at the theatre watching a show by the Kransky Sisters, and I can confirm that they are just as funny on stage as they are on the television (or, I suppose, on the wireless).
For those of you not familiar with the strange, morbid tone of the Kranskys, here's a sample. This is one of their older performances - Arva (the one playing the tuba) has since left the group. According to the other sisters, Arva received a scholarship to join the Hornbell Military Marching Band, and no longer tours with her sisters because "all the meals are included with the army" (and the camouflage uniforms hide all the spills). Fortunately, a fourth sister, Dawn, has left her job as a part-time trolley librarian in the local hospital to join the group. Dawn had been living in the neighbour's laundry, where she kept her collection of cane toad skins that she had flattened in the clothes-wringer.
- Home
- Angry by Choice
- Catalogue of Organisms
- Chinleana
- Doc Madhattan
- Games with Words
- Genomics, Medicine, and Pseudoscience
- History of Geology
- Moss Plants and More
- Pleiotropy
- Plektix
- RRResearch
- Skeptic Wonder
- The Culture of Chemistry
- The Curious Wavefunction
- The Phytophactor
- The View from a Microbiologist
- Variety of Life
Field of Science
-
-
Change of address9 months ago in Variety of Life
-
Change of address9 months ago in Catalogue of Organisms
-
-
Earth Day: Pogo and our responsibility1 year ago in Doc Madhattan
-
What I Read 20241 year ago in Angry by Choice
-
I've moved to Substack. Come join me there.1 year ago in Genomics, Medicine, and Pseudoscience
-
-
-
-
Histological Evidence of Trauma in Dicynodont Tusks7 years ago in Chinleana
-
Posted: July 21, 2018 at 03:03PM7 years ago in Field Notes
-
Why doesn't all the GTA get taken up?7 years ago in RRResearch
-
-
Harnessing innate immunity to cure HIV9 years ago in Rule of 6ix
-
-
-
-
-
-
post doc job opportunity on ribosome biochemistry!11 years ago in Protein Evolution and Other Musings
-
Blogging Microbes- Communicating Microbiology to Netizens11 years ago in Memoirs of a Defective Brain
-
Re-Blog: June Was 6th Warmest Globally11 years ago in The View from a Microbiologist
-
-
-
The Lure of the Obscure? Guest Post by Frank Stahl13 years ago in Sex, Genes & Evolution
-
-
Lab Rat Moving House14 years ago in Life of a Lab Rat
-
Goodbye FoS, thanks for all the laughs14 years ago in Disease Prone
-
-
Slideshow of NASA's Stardust-NExT Mission Comet Tempel 1 Flyby15 years ago in The Large Picture Blog
-
in The Biology Files
How to Recognise a Species

On a couple of previous occasions, I've discussed the use of the Biological Species Concept (BSC) versus the Phylogenetic Species Concept* (PSC) - see posts here and here, the first of which defines the two terms if you're not familiar with them. One question that someone, somewhere will always bring up whenever I broach the subject is whether use of the PSC would require the division of humans into separate species, so this post is partially in response to that. From a more general perspective, I thought that some of you might be interested simply in knowing how a taxonomist recognises if they have a new species.
*The more I cover this topic, though, the less I like the term "phylogenetic species concept", because the PSC isn't determined solely, or even primarily, by phylogeny. I prefer the term "diagnostic species concept". Real, honest-to-goodness phylogenetic species concepts, where species are defined by holophyly, have been proposed before (e.g. Donoghue, 1985) and I think the term should be reserved for them. Such concepts have never been particularly popular, though, because frankly they're shit.
The first point, that I think I've said before but can never be stressed enough, is that whatever species concept you are using, a species represents a hypothesis. Sadly, specimens do not come with convenient labels saying to what species they belong. If they did, I'd probably be out of a job (not that I won't soon be out of a job - that thesis deadline looks like it's getting closer every day*). A taxonomist presented with an array of specimens will divide them up into what appear to be distinct species, but that division will always be subject to further revision. Intermediate specimens may be found that blur the boundaries between previously distinct morphologies. Two different "species" may hatch out of eggs laid by a single parent. That sort of thing. No taxonomic revision can ever be final.
*Oh, wait...

What most taxonomists are looking for when distinguishing species are discrete, discontinuous characters. Whatever your favoured species concept, ultimately what you are trying to identify are populations that are genetically isolated from others. To give an example, Megalopsalis sp. A is found in Western Australia (I'm calling it "sp. A" here because 'tain't published yet). It differs from other species of Megalopsalis because the femur of the pedipalp (a little leg-like appendage in front of the legs that's used for feeding, visible in the photo below of Protolophus by Robert Pearson) has a row of spines running along the top (dorsal) edge. Over in Victoria and South Australia lives Megalopsalis hoggi, a closely related species. Specimens of M. hoggi never have spines on top of the pedipalp, but they may or may not have spines underneath it (ventrally).
You may be wondering why I regard presence or absence of dorsal spines as a character distinguishing species, but not presence or absence of ventral spines. After all, both are equally distinct. The difference is a matter of geography. In the specimens that I've seen of Megalopsalis sp. A, all of them have dorsal spines on the pedipalp, and I've not seen that character in Megalopsalis specimens outside the range of species A. Specimens with dorsal spines are geographically discontinuous from specimens without, indicating that they represent a distinct population. However, in the case of M. hoggi, specimens with ventral spines on the pedipalp are found in the same locality as specimens without ventral spines. The two are not discontinuous, suggesting that in this case that they are not genetically isolated.

It is possible that the ventrally-spined and ventrally-spineless individuals do represent separate species, but with overlapping distributions (and indeed, western specimens of M. hoggi tend to have spines while eastern specimens tend not to). If so, then study of other character complexes (such as population genetics) may reinforce their distinction. It is also possible that future collections may uncover overlapping variation in the Western Australian population that I've called "species A", and it may not be so distinct after all. But the recognition of two species, one variable and one not, is the most consistent with the information I have on hand so far.
Which leads up to my second point - pretty much any character could potentially distinguish a species, but pretty much any character could also potentially not distinguish a species. The ultimately important factor isn't the characters themselves - it's what they say about the gene flow leading to their current distribution. A single widespread species may encompass more variation within its distribution than a whole complex of closely-related species with restricted ranges. The difference is that character distributions will overlap in the former case but not in the latter.
So to get back to the question of whether use of the PSC would lead to recognition of separate species - personally, not having studied human variation to any great extent*, I don't actually know (really, it's exactly the same question as whether human races have any biological validity, just phrased using different terminology). I suspect that it wouldn't - while humans do vary considerably over their extraordinarily wide range, most of that variation is probably clinal rather than falling into discrete populations. It doesn't reflect underlying barriers to gene flow. This is especially the case in the modern world, where increased transportation and migration is breaking down what geographical barriers there may once have been.
*As organisms go, primates are mostly fairly dull.
As an unrelated postscript, in both of the earlier posts I've referred to a "tendency" of using the PSC to lead to recognition of more species than the BSC. Which got me thinking - are there any situations where the PSC would recognise less species than the BSC? Some possibilities that occured to me were situations involving polyploidy, or cases where interfertility was determined by the action of a single gene such as chirality reversals in gastropods. In these cases, diploid vs. polyploid or dextral vs. sinistral individuals might not be directly interfertile (which would mean they belonged to distinct biological species), but mutation from one state to the other might mean that there was still a reasonably steady gene flow going on (so they would not be distinct phylogenetic species). Any thoughts?
REFERENCES
Donoghue, M. J. (1985). A critique of the biological species concept and recommendations for a phylogenetic alternative. Bryologist 88: 172-181.
The Butterflies Get All the Glory (Taxon of the Week: Gelechioidea)

The Lepidoptera, butterflies and moths*, are one of the most familiar groups of insects, and have the enviable advantage of tending to receive a more favourable response from the general public than other insects do. Some Lepidoptera, particularly the butterflies, are almost treated as honorary vertebrates - they receive a degree of attention and enthusiasm usually reserved for those animals of a more endoskeletal disposition. You might be forgiven, then, for assuming that the Lepidoptera are overall a well-known and studied order - but you'd still be wrong.
*Though in our lab, they tend to get referred to simply as "leps".
Traditionally, Lepidoptera have been divided into two, reasonably self-explanatory, groups - the microlepidoptera and macrolepidoptera. Though originally divided simply by size, the terms have been redefined in recent years on a phylogenetic basis - the name Macrolepidoptera has been attached to a particular clade, while leps outside this clade are dubbed microlepidoptera. The intuitive meanings of the names still work reasonably well - the majority of large Lepidoptera are indeed Macrolepidoptera (though the often very sizeable Hepialidae are, phylogenetically speaking, microlepidoptera). And, not surprisingly, it is the Macrolepidoptera that get most of the attention, while the microleps (which, just to make the point, probably constitute the greater part of lepidopteran diversity) tend to get shoved to one side.

The Gelechioidea are a large microlepidopteran superfamily. They are spectacularly diverse - Hodges (1998) referred to there being well over 16,000 described species. As if this wasn't impressive enough, perhaps only about a quarter of the world's gelechioids have been described. Most of the Gelechioidea are extremely small - one of the largest, Cryptophasa setiotricha, is 25 mm long, while one of the smallest is Siskiwitia falcata, a mere two millimetres in length (Hodges, 1998). Like many other tiny moths, the wings often have a long fringe of hairs. The larvae of gelechioids are usually retiring herbivores, often concealing themselves in a web of silk or binding leaves to form a hide. Some are detritivores or fungivores, while a few have become carnivores of other insects such as scales (Kaila, 2004). The monophyly of the Gelechioidea is not certain - the morphological analysis of Kaila (2004) supported gelechioid monophyly, but three of the four supporting characters were homoplasious with other Lepidoptera, while the only character unique to Gelechioidea (antennae meeting mesally in the pupa) had been lost in a number of gelechioid subgroups.

Homoplasy was similarly rife within the superfamily itself. Kaila (2004) resolved two major clades, a "gelechiid lineage" including (among others) the Gelechiidae, Cosmopterigidae and Coleophoridae, and an "oecophorid lineage" with the Xyloryctidae, Oecophoridae and Elachistidae, but both were supported solely by homoplasious characters. Bucheli & Wenzel (2005) used molecular data as well as morphology, but included less taxa in their analysis than Kaila (2004) - they continued to support the oecophorid lineage, but resolved the gelechiid lineage as paraphyletic. Probably as a result of such rampant homoplasy, no two revisions have agreed on the best way to divide the gelechioids into families - the major families listed are fairly safe, but various minor "family-type groups" move in and out of them at will.
Relatively few gelechioids are of economic significance to humans. A few are plant pests (such as Pectinophora gossypiella, the cotton bollworm) or can feed on stored grain or textiles, but for the most part they are just as retiring as they look. There's still an awful lot of them, though.
REFERENCES
Bucheli, S. R., & J. Wenzel. 2005. Gelechioidea (Insecta: Lepidoptera) systematics: A reexamination using combined morphology and mitochondrial DNA data. Molecular Phylogenetics and Evolution 35 (2): 380-394.
Hodges, R.W., 1998. The Gelechioidea. In Lepidoptera: Moths and Butterflies (N. P. Kristensen, ed.) pp. 131–158. Walter de Gruyter, Berlin and New York.
Kaila, L. 2004. Phylogeny of the superfamily Gelechioidea (Lepidoptera: Ditrysia): an exemplar approach. Cladistics 20 (4): 303-340.
'Cause Every Night is New Year's Eve - One More Time!
Submissions are being called in again for this year's Open Lab collection. Last year, this site was represented - will it be so again this year?
So at any time until the end of the year, if you see a post here that looks particularly impressive, click on the button in the sidebar that looks like this:

and you'll be whisked away to a form where you can enter the details. You can submit any post that's been published since the 1st of December, 2008. Thank you for your support!
So at any time until the end of the year, if you see a post here that looks particularly impressive, click on the button in the sidebar that looks like this:

and you'll be whisked away to a form where you can enter the details. You can submit any post that's been published since the 1st of December, 2008. Thank you for your support!
It's Not What You Think
A little less than a year ago, I mentioned the strange and extremely cool phenomenon of independent gametophytes in ferns - cases where the tiny haploid gametophyte generation of a fern is able to reproduce asexually and hang around as a plant that, to the untrained eye, wouldn't look much like a fern at all. In that post, I said that independent gametophytes were known for "a single species of Grammitidaceae, two Vittariaceae and nine Hymenophyllaceae". A paper just out in Plant Systematics and Evolution (Li et al., 2009) identifies another independent gametophyte - and this is the most mind-blowing of all. Not only does it come from a family for which independent gametophytes have not previously been recorded, but it turns out to have been hiding in very plain view.

This is it! (Photo from here.)
Süßwassertang (or "suesswassertang") is a plant that people in Europe and North America have been growing in their aquaria for a few years now (the name is German for "freshwater seaweed"). Specimens are propagated vegetatively by simply breaking them apart. I haven't been able to find out exactly where it originally came from - internet fora refer to a probable source from a German botanic garden, but it seems that specimens have mostly been passed around by private individuals (see this discussion, for instance). The original assumption seems to have been that it was some sort of liverwort, like a similar-looking aquarium plant known as Monosolenium tenerum or Pellia* (in fact, Süßwassertang has also been referred to as "round pellia", in reference to its different growth habit from true pellia). However, rarely produced gametangia (reproductive organs) suggested that it may be a fern gametophyte instead, and this has been confirmed by Li et al. through molecular analysis.
*It originally appeared on the market as Pellia, but has since been re-designated Monosolenium tenerum (see here). The Wikipedia page on Monosolenium suggests that this may also be wrong, but doesn't give any sources for this claim. Liverworts are far from easy to identify, so it's not outside the realms of possibility.
Süßwassertang turns out to be very closely related to Lomariopsis lineata in the Lomariopsidaceae, which looks like this (photo by Julie Barcelona):

Lomariopsis lineata is an Asian species of the pantropical epiphytic fern genus Lomariopsis, members of which can climb up trees on long running stems to heights of ten metres (Rouhan et al., 2007). That an arboreal fern could produce an independent gametophyte is surprising - that such a gametophyte should be aquatic is incredible. A number of websites have already started referring to Süßwassertang as Lomariopsis lineata, but this is jumping the gun a little. To date, no Süßwassertang specimens have been successfully induced to produce sporophytes, despite their occassional production of gametangia (normally in ferns, gametophytes produce male and female gametangia, the gametes from which fertilise each other and grow into sporophytes). Even when Süßwassertang were transplanted into terrestrial conditions, no sporophytes were produced though gametangia production increased (on the other hand, their growth was much reduced). Süßwassertang has so far only been demonstrated to be extremely close to L. lineata, not necessarily identical with it.
As alluded to in the previous post, independent gametophytes may be able to survive in conditions which their relevant sporophytes would find intolerable. The Süßwassertang would seem to be one of the ultimate examples.
REFERENCES
Li, F.-W., B. C. Tan, V. Buchbender, R. C. Moran, G. Rouhan, C.-N. Wang & D. Quandt. 2009. Identifying a mysterious aquatic fern gametophyte. Plant Systematics and Evolution 281 (1): 77-86.
Rouhan, G., J. G. Hanks, D. McClelland & R. C. Moran. 2007. Preliminary phylogenetic analysis of the fern genus Lomariopsis (Lomariopsidaceae). Brittonia 59 (2): 115-128.

Süßwassertang (or "suesswassertang") is a plant that people in Europe and North America have been growing in their aquaria for a few years now (the name is German for "freshwater seaweed"). Specimens are propagated vegetatively by simply breaking them apart. I haven't been able to find out exactly where it originally came from - internet fora refer to a probable source from a German botanic garden, but it seems that specimens have mostly been passed around by private individuals (see this discussion, for instance). The original assumption seems to have been that it was some sort of liverwort, like a similar-looking aquarium plant known as Monosolenium tenerum or Pellia* (in fact, Süßwassertang has also been referred to as "round pellia", in reference to its different growth habit from true pellia). However, rarely produced gametangia (reproductive organs) suggested that it may be a fern gametophyte instead, and this has been confirmed by Li et al. through molecular analysis.
*It originally appeared on the market as Pellia, but has since been re-designated Monosolenium tenerum (see here). The Wikipedia page on Monosolenium suggests that this may also be wrong, but doesn't give any sources for this claim. Liverworts are far from easy to identify, so it's not outside the realms of possibility.
Süßwassertang turns out to be very closely related to Lomariopsis lineata in the Lomariopsidaceae, which looks like this (photo by Julie Barcelona):

Lomariopsis lineata is an Asian species of the pantropical epiphytic fern genus Lomariopsis, members of which can climb up trees on long running stems to heights of ten metres (Rouhan et al., 2007). That an arboreal fern could produce an independent gametophyte is surprising - that such a gametophyte should be aquatic is incredible. A number of websites have already started referring to Süßwassertang as Lomariopsis lineata, but this is jumping the gun a little. To date, no Süßwassertang specimens have been successfully induced to produce sporophytes, despite their occassional production of gametangia (normally in ferns, gametophytes produce male and female gametangia, the gametes from which fertilise each other and grow into sporophytes). Even when Süßwassertang were transplanted into terrestrial conditions, no sporophytes were produced though gametangia production increased (on the other hand, their growth was much reduced). Süßwassertang has so far only been demonstrated to be extremely close to L. lineata, not necessarily identical with it.
As alluded to in the previous post, independent gametophytes may be able to survive in conditions which their relevant sporophytes would find intolerable. The Süßwassertang would seem to be one of the ultimate examples.
REFERENCES
Li, F.-W., B. C. Tan, V. Buchbender, R. C. Moran, G. Rouhan, C.-N. Wang & D. Quandt. 2009. Identifying a mysterious aquatic fern gametophyte. Plant Systematics and Evolution 281 (1): 77-86.
Rouhan, G., J. G. Hanks, D. McClelland & R. C. Moran. 2007. Preliminary phylogenetic analysis of the fern genus Lomariopsis (Lomariopsidaceae). Brittonia 59 (2): 115-128.
More Gagrellinae (Taxon of the Week: Harmanda)
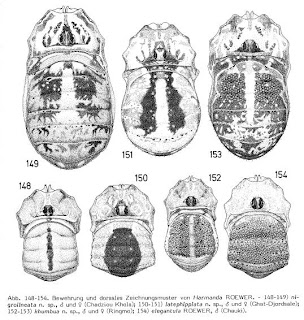
I've written before about the horrors attending on the Gagrellinae, a subfamily of mostly tropical harvestmen found in Asia and the Americas. But it's not all doom and gloom - outside the black hole that is the Gagrella concept, there's some sections that are quite pleasant.
Harmanda is one of the safer gagrelline genera, because unlike most gagrellines it's actually been revised, by Jochen Martens in 1987. Martens listed eleven species (one with four subspecies) from the Himalayas in the genus united by their distinctive male genital morphology - very flat, relatively broad shaft, with the mantle "wings" starting very shortly after the glans, and a long muscle extending at least half the length of the glans. Harmanda species also possess a short, round apophysis (side-branch) on the pedipalp patella (look at the pictures if you want to know what I mean - offhand, the pictures I'm using today are taken from Martens, 1987, because online images of Harmanda seem to be non-existent). Martens didn't mention two species earlier included in the genus by Roewer (1955) - Harmanda annulata from northern India and H. triseriata from Singapore. Like other gagrelline genera, Roewer assigned species to Harmanda based on little more than the number of nodules in each leg, a character that may not even be constant within one individual, let alone one genus, and the isolated geographic location of the Singapore species at least suggests that it may not be Harmanda. Roewer (1954)also divided Asian Gagrellinae into two "groups", Gagrelleae and Zalepteae, based on the presence (Gagrelleae) or absence (Zalepteae) of large central spines on the abdomen, and Harmanda would be zaleptean by this measure. But again, this feature was probably over-rated by Roewer - Martens (1987) described one genus, Pokhara, whose members were clearly related on the basis of genital morphology, but which included both "gagrellean" and "zaleptean" species.

Martens (1987) commented that Harmanda included some of the most eye-catching Himalayan Gagrellinae, with brightly metallic-coloured abdomens (metallic colours are unusual among arachnids, but a number of Gagrellinae go in for them). Most are inhabitants of oak and conifer forests, and can reach reasonable altitudes - Harmanda medioimmicans gets up to 3600 m.
Finally, I'll point out that northern India and the Himalayas may be a significant centre of diversity for Gagrellinae - a decent proportion of the species in this subfamily have been described from there. But the obvious question is, is this area truly particularly speciose, or have the harvestmen there been better studied than elsewhere?
REFERENCES
Martens, J. 1987. Opiliones aus dem Nepal-Himalaya VI. Gagrellinae (Arachnida: Phalangiidae). Courier Forschungsinstitut Senckenberg 93: 87-202.
Roewer, C. F. 1954. Indoaustralische Gagrellinae (Opiliones, Arachnidae). (Weitere Weberknechte XVIII). 1. Teil. Senckenbergiana Biologica 35 (3-4): 181-236.
Roewer, C. F. 1955. Indoaustralische Gagrellinae (Opiliones, Arachnidae). (Weitere Weberknechte XVIII). 4. Teil (Schluß). Senckenbergiana Biologica 36 (3-4): 123-171.
I Can't Believe I'm Falling For A Meme
...but here are the rules, as passed on by Greg Laden:
Except here's the thing: one of the first books that came to mind was one that I can't even remember the title of. It was a huge book (well, huge to a smallish child) that the local library had when I was about ten, probably on South American wildlife, but I just remember these huge double-page dioramas of such things as South American palaeofauna - litopterns, notoungulates and astrapotheres, oh my! - or army ants on the move. Like I said, wouldn't have a clue what it was called now. I'm not even sure that I knew it at the time - it was just the really big book with the cool pictures of Macrauchenia. Similar issues of memory surround that big book on birds that I used to spend hours looking at every time I visited my great-grandmother.
So, if I obmit those ones, here's my list:
1 - Pride and Prejudice by Jane Austen. Yes, it had to be in there somewhere. And to follow on from an earlier meme, I can now say that I've read all the Austen novels. Pride and Prejudice isn't necessarily the best - I'd say that that title probably belongs to Persuasion, while Northanger Abbey deserves a lot more credit than it's usually given, but I will admit that P & P is probably the most memorable.
2 - Peace on Earth by Stanislaw Lem. It was a toss-up between this one and Solaris. I suspect that only a small proportion of Lem's books have been translated into English, and those that have been are scattered far and wide and almost impossible to find, but every time you do find one you can feel guaranteed that it's an absolute gem. I'd also recommend The Cyberiad.
3 - Slaughterhouse Five by Kurt Vonnegut. Again, with an honourable mention to Galapagos.
4 - The Kuia and the Spider by Patricia Grace. I cited this children's book in an earlier post. What I didn't mention there was that The Kuia and the Spider was actually a revolutionary experience for me as a young child, and one that I'll confess I didn't appreciate right away - it was the first time I was introduced to the idea that a story could have an ending without having a conclusion.
5 - Titus Groan by Mervyn Peake. I read this book a lot when I was in high school. It still shows through every time I write something.
6 - The Old Man and the Sea by Ernest Hemingway. I hated this book. We had to read it in high school - and watch the stoopid film adaptation. Bored me to tears. But you know what - almost everything else we read in high school English is long forgotten, but I don't think I'm ever going to forget Hemingway's musings on the true natures of triumph and futility.
7 - The Book of Lost Tales by J. R. R. Tolkein. I'm going to say something that will probably result in my geek status being instantly revoked - Tolkein's not really that great. But there's something about the experience of Tolkein, the whole idea of all the hours of scholarship hidden behind the finished product, that somehow ends up imbuing the whole with something far, far more than the cardboard characters, clunky prose and abominable poetry. The Book of Lost Tales, a collection of early versions of what would end up becoming The Silmarillion (and, offhand, written in a more paganistic style than the more Catholic morality of the later work) are something of a glimpse behind the magician's curtain. And the dragons are mechanical.
8 - Die Weberknechte der Erde: Systematisches Bearbeitung der Bisher Bekannten Opiliones by Carl-Friedrich Roewer. Again, I'll never forget it for all the wrong reasons. I don't know that I can say I've really read "Die Weberknechte" - I don't think that even people who actually speak German can understand Roewer. But just try typing "Roewer" into the search box for this blog to get some idea of how much his shadow hangs over my professional career.
9 - The Hitch-hiker's Guide to the Galaxy by Douglas Adams. What more need I say?
10 - Perelandra by C. S. Lewis. With the benefit of hindsight and greater maturity, I know that it's as subtle as a half-brick. But some bits of this book genuinely creeped me out when I was a kid.
11 - Hard-Boiled Wonderland and the End of the World by Haruki Murakami. I got handed this book to read as part of my Japanese Culture class when I was in Fukuoka. I think that by the end of the day, I'd been through it twice. And it's the only work of popular fiction I've known to reference Synthetoceras and Cranioceras (even if the process of re-translating a Japanese transliteration of the original names made them all but unrecognisable - I think Cranioceras became "kuranokeras").
12 - Alice Through the Looking-Glass by Lewis Carroll. Together with Alice's Adventures in Wonderland, and bits (but not all) of the Sylvie and Bruno books, I couldn't get enough Carroll.
13 - Paradise Lost by John Milton. You always remember Paradise Lost. Milton later tried to remove the Devil's triumph by writing Paradise Regained. He failed.
14 - The Blue Mountain by Meir Shalev. Are the stories true? Are they lies? Does it really matter?
15 - The Just-So Stories by Rudyard Kipling. Finally, one of the most memorable of all. "He was wild and he was woolly, and his pride was inordinate, and he danced on an outcrop in the middle of Australia". "I am the cat who walks by himself, and all places are alike to me". "The great grey-green Limpopo river, all set about with fever trees". The inimatable rhythm of Kipling's stories is truly unforgettable.
Don't take too long to think about it. Fifteen books you've read that will always stick with you.
First 15 you can recall in no more than 15 minutes.
Except here's the thing: one of the first books that came to mind was one that I can't even remember the title of. It was a huge book (well, huge to a smallish child) that the local library had when I was about ten, probably on South American wildlife, but I just remember these huge double-page dioramas of such things as South American palaeofauna - litopterns, notoungulates and astrapotheres, oh my! - or army ants on the move. Like I said, wouldn't have a clue what it was called now. I'm not even sure that I knew it at the time - it was just the really big book with the cool pictures of Macrauchenia. Similar issues of memory surround that big book on birds that I used to spend hours looking at every time I visited my great-grandmother.
So, if I obmit those ones, here's my list:
1 - Pride and Prejudice by Jane Austen. Yes, it had to be in there somewhere. And to follow on from an earlier meme, I can now say that I've read all the Austen novels. Pride and Prejudice isn't necessarily the best - I'd say that that title probably belongs to Persuasion, while Northanger Abbey deserves a lot more credit than it's usually given, but I will admit that P & P is probably the most memorable.
2 - Peace on Earth by Stanislaw Lem. It was a toss-up between this one and Solaris. I suspect that only a small proportion of Lem's books have been translated into English, and those that have been are scattered far and wide and almost impossible to find, but every time you do find one you can feel guaranteed that it's an absolute gem. I'd also recommend The Cyberiad.
3 - Slaughterhouse Five by Kurt Vonnegut. Again, with an honourable mention to Galapagos.
4 - The Kuia and the Spider by Patricia Grace. I cited this children's book in an earlier post. What I didn't mention there was that The Kuia and the Spider was actually a revolutionary experience for me as a young child, and one that I'll confess I didn't appreciate right away - it was the first time I was introduced to the idea that a story could have an ending without having a conclusion.
5 - Titus Groan by Mervyn Peake. I read this book a lot when I was in high school. It still shows through every time I write something.
6 - The Old Man and the Sea by Ernest Hemingway. I hated this book. We had to read it in high school - and watch the stoopid film adaptation. Bored me to tears. But you know what - almost everything else we read in high school English is long forgotten, but I don't think I'm ever going to forget Hemingway's musings on the true natures of triumph and futility.
7 - The Book of Lost Tales by J. R. R. Tolkein. I'm going to say something that will probably result in my geek status being instantly revoked - Tolkein's not really that great. But there's something about the experience of Tolkein, the whole idea of all the hours of scholarship hidden behind the finished product, that somehow ends up imbuing the whole with something far, far more than the cardboard characters, clunky prose and abominable poetry. The Book of Lost Tales, a collection of early versions of what would end up becoming The Silmarillion (and, offhand, written in a more paganistic style than the more Catholic morality of the later work) are something of a glimpse behind the magician's curtain. And the dragons are mechanical.
8 - Die Weberknechte der Erde: Systematisches Bearbeitung der Bisher Bekannten Opiliones by Carl-Friedrich Roewer. Again, I'll never forget it for all the wrong reasons. I don't know that I can say I've really read "Die Weberknechte" - I don't think that even people who actually speak German can understand Roewer. But just try typing "Roewer" into the search box for this blog to get some idea of how much his shadow hangs over my professional career.
9 - The Hitch-hiker's Guide to the Galaxy by Douglas Adams. What more need I say?
10 - Perelandra by C. S. Lewis. With the benefit of hindsight and greater maturity, I know that it's as subtle as a half-brick. But some bits of this book genuinely creeped me out when I was a kid.
11 - Hard-Boiled Wonderland and the End of the World by Haruki Murakami. I got handed this book to read as part of my Japanese Culture class when I was in Fukuoka. I think that by the end of the day, I'd been through it twice. And it's the only work of popular fiction I've known to reference Synthetoceras and Cranioceras (even if the process of re-translating a Japanese transliteration of the original names made them all but unrecognisable - I think Cranioceras became "kuranokeras").
12 - Alice Through the Looking-Glass by Lewis Carroll. Together with Alice's Adventures in Wonderland, and bits (but not all) of the Sylvie and Bruno books, I couldn't get enough Carroll.
13 - Paradise Lost by John Milton. You always remember Paradise Lost. Milton later tried to remove the Devil's triumph by writing Paradise Regained. He failed.
14 - The Blue Mountain by Meir Shalev. Are the stories true? Are they lies? Does it really matter?
15 - The Just-So Stories by Rudyard Kipling. Finally, one of the most memorable of all. "He was wild and he was woolly, and his pride was inordinate, and he danced on an outcrop in the middle of Australia". "I am the cat who walks by himself, and all places are alike to me". "The great grey-green Limpopo river, all set about with fever trees". The inimatable rhythm of Kipling's stories is truly unforgettable.
Request For a Citation
The recently-published (well, reasonably recently) 2008 edition of The Open Laboratory included a copy of an article by yours truly. Unfortunately, due to a number of factors (mostly due to being a destitute student who, at present, doesn't know whether his scholarship is going to be extended past the end of the month), I haven't yet been able to afford my own copy of the book, but I would like to be able to cite it on my CV. So if anyone out there does have a copy, would you be able to flick to the table of contents and tell me what pages I'm on? Thank you.
A Minor Complaint about Google, and a Major Complaint about Ranked Taxonomy
There's a couple of things bugging me today, so please forgive me if I take the opportunity to vent (albeit in as genteel a manner as I can).
First, what has happened to the "web search" option on Google Scholar? Google Scholar is a fantastic resource, and one that has made itself indispensable to my life, so I'm certainly not complaining about Scholar as a whole. But until recently, each of the reference search results that came back would have a "web search" link among the options beneath it, which if clicked would automatically search for that reference on ordinary Google. This came in handy on a regular basis - often an online and/or free copy of the article might not be found by Scholar, but would be found by Google Normal (for instance, if the copy was hosted by a researcher's personal webpage). But just recently that option seems to have vanished, meaning I have to cut and past results from Scholar to Google myself. Grumble, grumble, they're stealing seconds from my life...
The second thing that's bugging me is a little more significant than a moment's inconvenience in searching, though (at least, I think it is, and it's my blog after all). My thesis is rapidly approaching completion (or to be more accurate, perhaps, rapidly approaching deadline) and that includes the big phylogenetic analysis of Monoscutidae. One of the problem areas in this family involves the previously poorly-defined and heterogeneous genus Spinicrus, and recently (in Taylor & Hunt, 2009) I segregated one of the more distinctive species (plus some previously unpublished species) as part of the new genus Neopantopsalis. I hadn't yet done any of the phylogenetic analysis at that time - rather, Neopantopsalis was established purely on the basis of its distinctive characters.
Fast-forward to now, when the greater part of the analysis is taking shape, and one interesting result is that the greater part of the genus Spinicrus looks like it belongs to a single clade after all, with Neopantopsalis nested within that clade. The relevant part of the tree currently looks like this:
0--Spinicrus tasmanicum
`--+--Spinicrus stewarti
`--+--+--Spinicrus minimum
| `--Spinicrus porongorupense
`--Neopantopsalis (5 spp.)
In other words, Spinicrus minus Neopantopsalis is paraphyletic. Neopantopsalis itself is still well-supported as a clade (more so than the Spinicrus clade as a whole, in fact), and from a diagnostic perspective the differences between the remaining Spinicrus species are less obvious than the differences between any of them and Neopantopsalis. So these are my options:
Of course, I still have a couple of species left to include in the analysis, and it's possible that once they're in there, Neopantopsalis might move out of Spinicrus and the whole problem will go away. But let's assume for now that it doesn't.
The punchline to all this, which I'm sure some of you have realised long before now, is that it is only the use of a ranked taxonomic system, and hence my need to decide which clade(s) I'll recognise at the rank of "genus", that even makes this a problem in the first place. If I was dealing with an entirely unranked system, I could simply recognise a clade Neopantopsalis, positioned within a larger clade Spinicrus, and that would be an end to it. I could go ahead and use a genus-free taxonomy as has been done by some authors, but at the present time that would almost guarantee that no-one uses my nomenclature - not the result I'm going for. So I sit here, forced to mull over the pros and cons about how to solve a problem that, really, shouldn't even be a problem at all.
REFERENCES
Taylor, C. K., & G. S. Hunt. 2009. New genus of Megalopsalidinae (Arachnida: Opiliones: Monoscutidae) from north-eastern Australia. Zootaxa 2130: 41-59.
First, what has happened to the "web search" option on Google Scholar? Google Scholar is a fantastic resource, and one that has made itself indispensable to my life, so I'm certainly not complaining about Scholar as a whole. But until recently, each of the reference search results that came back would have a "web search" link among the options beneath it, which if clicked would automatically search for that reference on ordinary Google. This came in handy on a regular basis - often an online and/or free copy of the article might not be found by Scholar, but would be found by Google Normal (for instance, if the copy was hosted by a researcher's personal webpage). But just recently that option seems to have vanished, meaning I have to cut and past results from Scholar to Google myself. Grumble, grumble, they're stealing seconds from my life...
The second thing that's bugging me is a little more significant than a moment's inconvenience in searching, though (at least, I think it is, and it's my blog after all). My thesis is rapidly approaching completion (or to be more accurate, perhaps, rapidly approaching deadline) and that includes the big phylogenetic analysis of Monoscutidae. One of the problem areas in this family involves the previously poorly-defined and heterogeneous genus Spinicrus, and recently (in Taylor & Hunt, 2009) I segregated one of the more distinctive species (plus some previously unpublished species) as part of the new genus Neopantopsalis. I hadn't yet done any of the phylogenetic analysis at that time - rather, Neopantopsalis was established purely on the basis of its distinctive characters.
Fast-forward to now, when the greater part of the analysis is taking shape, and one interesting result is that the greater part of the genus Spinicrus looks like it belongs to a single clade after all, with Neopantopsalis nested within that clade. The relevant part of the tree currently looks like this:
0--Spinicrus tasmanicum
`--+--Spinicrus stewarti
`--+--+--Spinicrus minimum
| `--Spinicrus porongorupense
`--Neopantopsalis (5 spp.)
In other words, Spinicrus minus Neopantopsalis is paraphyletic. Neopantopsalis itself is still well-supported as a clade (more so than the Spinicrus clade as a whole, in fact), and from a diagnostic perspective the differences between the remaining Spinicrus species are less obvious than the differences between any of them and Neopantopsalis. So these are my options:
- Sink Neopantopsalis back into Spinicrus: This has the advantage of re-instating Spinicrus as a monophyletic unit, but disguises the distinctiveness of the 'Neopantopsalis' group. It also means that Spinicrus is back to being horribly heterogeneous again.
- Retain Spinicrus as a paraphyletic grouping, excluding Neopantopsalis as a separate genus: I'm not entirely prejudiced against paraphyla when they appear to be useful, and I do regard this as a viable option diagnostically. However, it disguises the point that some Spinicrus are closer to Neopantopsalis than others, and I would remind you all that monophyly-based classifications are likely to have more predictive power in the long run.
- Divide Spinicrus into a number of genera - one for each successive outgroup to Neopantopsalis: This manages to both recognise the distinctiveness of Neopantopsalis and avoid paraphyla, but it's not an option I find appealing at present. The problem is that the diagnostic characters for the other ex-Spinicrus isolates would probably end up being fine scale characters such as spiracle ultrastructure and the appearance of the sensory pores on the genitalia. Significant characters phylogenetically, but very difficult (if not impossible) to distinguish without the use of an electron microscope. When it comes to working classifications, I am sympathetic to the view that practicality trumps strict accuracy.
- Synonymise Neopantopsalis with Spinicrus, but as a distinct subgenus: This is the option that is currently appealing to me the most - Neopantopsalis remains distinguished, and Spinicrus is still monophyletic. However, there are two potential complaints I can see. One is that, really, who uses subgenera anyway? The second is that I wouldn't establish subgenera for the other species (for the same reasons as I wouldn't establish separate genera for them), and I can imagine reviewers complaining about some species being placed to subgenus while others are not.
Of course, I still have a couple of species left to include in the analysis, and it's possible that once they're in there, Neopantopsalis might move out of Spinicrus and the whole problem will go away. But let's assume for now that it doesn't.
The punchline to all this, which I'm sure some of you have realised long before now, is that it is only the use of a ranked taxonomic system, and hence my need to decide which clade(s) I'll recognise at the rank of "genus", that even makes this a problem in the first place. If I was dealing with an entirely unranked system, I could simply recognise a clade Neopantopsalis, positioned within a larger clade Spinicrus, and that would be an end to it. I could go ahead and use a genus-free taxonomy as has been done by some authors, but at the present time that would almost guarantee that no-one uses my nomenclature - not the result I'm going for. So I sit here, forced to mull over the pros and cons about how to solve a problem that, really, shouldn't even be a problem at all.
REFERENCES
Taylor, C. K., & G. S. Hunt. 2009. New genus of Megalopsalidinae (Arachnida: Opiliones: Monoscutidae) from north-eastern Australia. Zootaxa 2130: 41-59.
Algal Threads (Taxon of the Week: Myrionemataceae)

The brown algae of the Phaeophyceae* are one of the predominant groups of multicellular algae in the modern ocean. Though not the most speciose of the three (that title belongs to the red Macrorhodophytina), they are in many places the most ecologically significant, as this is the group that includes the gigantic kelps that predominate and shape many parts of the marine environment. As significant as kelps are, though, they are not my focus for today. Instead, I'm going to look at a much smaller group of brown algae, the Myrionemataceae.
*The names "Phaeophyceae" or "Phaeophyta" have usually been used to refer to the monophyletic clade of multicellular brown algae. Cavalier-Smith & Chao (2006) recently proposed an expanded "class Phaeophyceae" to include the unicellular Phaeothamniophyceae and the very-basically-multicellular Schizocladia, with the traditional Phaeophyceae referred to as "subclass Melanophycidae". As Cavalier-Smith & Chao did not dispute the monophyly of the traditional Phaeophyceae, this is a simple ranking issue and I suspect it will probably be largely ignored by everyone else.
Myrionemataceae are minute filamentous algae, usually only a couple of millimetres in size (Abbott & Hollenberg, 1993). They generally grow in a disc only one or two cells deep, with numerous filaments growing upwards from the strands of the disc. These filaments may be uniserial or branching; they may or may not carry lateral processes or sporangia. Myrionemataceae most commonly seem to be epiphytic on much larger marine plants and algae.

Myrionemataceae (and small filamentous brown algae in genera) have been a royal pain in the neck for algal taxonomists, and as a result they have been widely shunned. For instance, the main distinguishing characteristic between the Myrionemataceae and the similarly filamentous Ectocarpaceae has been that the former is discoidal and the latter is not (Clayton, 1974). However, the genus Hecatonema (included by some authors in Myrionemataceae and excluded by others) includes some species that always grow in a disc, and others that do not. Some "Myrionemataceae" have been suggested to represent divergent growth forms or dimorphic generations of other brown algae placed in different families (many marine algae have very distinct alternating generations). This seems very probable for some species, but proving such relationships is not easy and Clayton (1972) claimed that at least some cases of myrionematoid algae being grown from cultures of other algae represented contamination instead of relationship. Other Myrionemataceae that have been cultured show no signs of alternating generations, while yet others have only ever been recorded reproducing asexually.

It seems almost certain that the "Myrionemataceae" are a polyphyletic grouping, with their overall lack of distinguishing features obscuring a diversity of origins. Peters (2003) found the type genus, Myrionema, to be itself polyphyletic. The type species of Myrionema, M. strangulans, has a fairly isolated position within the Ectocarpales sensu lato in Draisma et al. (2001), but it is pretty much anyone's guess as to how many of the so far unanalysed myrionematoid species belong with it*. Some species may be established as growth-forms of other taxa and shipped off accordingly, but others may prove more intractable and remain in the "family" - not so much a wastebasket as an inedible core.
*Das et al. (2005), in a paper that had me completely baffled but which I rather suspect was complete twaddle to begin with, cite a later phylogeny which they attribute to Draisma et al. (2002) that positions Myrionema strangulans in the Chordariaceae - unfortunately, Draisma et al. (2002) has absolutely no mention anywhere of Myrionema, chordariaceous or otherwise, so I wouldn't know what reference Das et al. really had in mind.
REFERENCES
Abbott, I. A., & G. J. Hollenberg. 1993. Marine Algae of California. Stanford University Press.
Cavalier-Smith, T., & E. E.-Y. Chao. 2006. Phylogeny and megasystematics of phagotrophic heterokonts (kingdom Chromista). Journal of Molecular Evolution 62: 388-420.
Clayton, M. N. 1972. The occurrence of variant forms in cultures of species of Ectocarpus and Giffordia. Br. phycol. J. 7: 101-108.
Clayton, M. N. 1974. Studies on the development, life history and taxonomy of the Ectocarpales (Phaeophyta) in southern Australia. Australian Journal of Botany 22: 743-813.
Das, S., J. Chakrabarti, Z. Ghosh, S. Sahoo & B. Mallick. 2005. A new measure to study phylogenetic relations in the brown algal order Ectocarpales: The “codon impact parameter”. J. Biosci. 30: 699–709.
Draisma, S. G. A., J. L. Olsen, W. T. Stam & W. F. Prud'homme van Reine. 2002. Phylogenetic relationships within the Sphacelariales (Phaeophyceae): rbcL, RUBISCO spacer and morphology. European Journal of Phycology 37: 385-401.
Draisma, S. G. A., W. F. Prud'homme van Reine, W. T. Stanm & J. L. Olsen. 2001. A reassessment of phylogenetic relationships within the Phaeophyceae based on RUBISCO large subunit and ribosomal DNA sequences. Journal of Phycology 37: 586-603.
Peters, A. F. 2003. Molecular identification, distribution and taxonomy of brown algal endophytes, with emphasis on species from Antarctica. In Seventeenth International Seaweed Symposium (A. R. O. Chapman, R. J. Anderson, V. J. Vreeland & I. R. Davison, eds) pp. 293-302. Oxford University Press.
Taşkin, E., M. Öztürk & M. J. Wynne. 2006. First report of Microspongium globosum Reinke (Phaeophyceae, Myrionemataceae) in the Mediterranean Sea. Nova Hedwigia 82 (1-2): 135-142.
Before the Word for World was Forest
...though to be perfectly honest, I've never read The Word for World is Forest, I just thought that it'd supply a catchy name for this post.

The environment of the Devonian Rhynie Chert, as illustrated by Zdenek Burian (via Palaeos.com.
If you want to find out about the evolution of terrestrial life for vertebrates, there are countless sources out there for you to turn to. But if you want to find out about the evolution of terrestrial life for plants, then your options are probably much thinner. Which is just one more example of how screwed up our priorities as humans are, because there's no doubt which is the greater achievement. When the first terrestrial vertebrates emerged, they found a world already made lush by a covering of vegetation. But the first terrestrial plants would have found nothing waiting for them but bare, hostile rock*. It's amazing that they ever managed at all.
*To be honest, I lie slightly. In places where there was available moisture, I'm sure that a film of bacteria would have grown. Ditto for unicellular algae and other such organisms. If lichen-type associations were around at the time (and a cyanobacteria-zygomycete association is preserved in the Rhynie Chert - Selosse & Le Tacon, 1998), the world would have been their mollusc. Sadly, with little potential for their fossilisation and discovery, we may never really know about the contributions of these first unicellular pioneers.

Reconstruction of the trimerophyte Psilophyton, from here.
But manage they did, and by the early Devonian the world was home to a small but respectable diversity of land plants. Most of the vascular plants of the time have been divided between the rhyniophytes, lycophytes, trimerophytes and cladoxylopsids (doubtless there were also moss- and liverwort-like plants around too, if not actual mosses and liverworts, but the spotty fossil record of bryophyte-grade plants doesn't quite reach that far back). Almost all of them, admittedly, would have been fairly similar to the non-expert eye - small, shrubby affairs with simple branching systems and no true leaves or roots. Examination of their fine structure (particularly of their vascular systems) is necessary to recognise their true affinities - rhyniophytes in the stem lineage for all vascular plants; lycophytes including the ancestors of modern Lycopodium, Selaginella and Isoetes; trimerophytes on the stem leading to modern ferns and seed plants; and cladoxylopsids on the stem of modern ferns*. Each of these groups quite possibly represents a grade rather than a clade, but in most cases it is not possible to actually demonstrate this one way or another.
*It is worth noting (especially in relation to a question asked in the comments to a previous post) that the vascular cells of rhyniophytes, lycophytes and trimerophytes each have distinct morphologies from each other** (Friedman & Cook, 2000), and this has led some authors to suggest that the vascular system may have developed independently in each of the three lineages. For now, though, it seems more parsimonious to assume a common origin followed by evolutionary divergence.
**It is also worth noting that when Friedman & Cook (2000) wrote their review, we actually knew more about the structure of the vascular cells in Devonian lycophytes and trimerophytes than in living lycophytes and ferns. Previous studies of vascular cell structure in living plants had almost exclusively looked at seed plants alone.
Trimerophytes differed from the more basal rhyniophytes in their mode of branching - whereas the basalmost land plants had branched dichotomously (dividing into two branches with each branch growing equivalently), trimerophytes branched anisotomously (one branch growing more than the other), effectively giving the trimerophytes some degree of a central stem (this process is called overtopping). Secondary branches from the central stem still branched dichotomously. Sporangia were borne on the tips of the branches, and at least some trimerophytes grew elongate sporangia in pairs that twisted around each other (Gerrienne, 1997).
 One particular trimerophyte, Psilophyton princeps, holds a particular significance for palaeobotany as the first Devonian plant to be reconstructed, by William Dawson in 1859 (Taylor & Krings, 2008), with a large creeping rhizome extending successive upright shoots. But perhaps even more significant was the size reached by some trimerophytes. While most Devonian vascular plants would have been struggling to reach half a metre in height, the trimerophyte Pertica dalhousii has been estimated to have reached up to three metres (Mauseth, 2008) - about the height of the ceiling of an average house (the related but smaller species Pertica quadrifaria is shown to the left, in a reconstruction from the Maine Geological Survey). Together with the similarly-sized cladoxylopsid Pseudosporochnus, these were effectively the first trees - not much compared to their modern successors, perhaps, but very impressive compared to anything that came before them (with the exception, of course, of the primordial oddity Prototaxites). It is interesting to imagine what the environment of these early "forests" would have been like. How did they handle the weather, for a start? In the absence of a strong root system to anchor them down, were they prone to collapsing in the wind? If this was so, did they grow rapidly to compensate for their short lives, or did the rhizome readily send up new shoots to replace lost ones? (Remember, with no leaves either, the entire stem would have probably been photosynthetic.) How did this affect life for the early terrestrial animals taking advantage of their presence? There may have been the beginnings of a forest, but a world recognisably our own was still a long way off.
One particular trimerophyte, Psilophyton princeps, holds a particular significance for palaeobotany as the first Devonian plant to be reconstructed, by William Dawson in 1859 (Taylor & Krings, 2008), with a large creeping rhizome extending successive upright shoots. But perhaps even more significant was the size reached by some trimerophytes. While most Devonian vascular plants would have been struggling to reach half a metre in height, the trimerophyte Pertica dalhousii has been estimated to have reached up to three metres (Mauseth, 2008) - about the height of the ceiling of an average house (the related but smaller species Pertica quadrifaria is shown to the left, in a reconstruction from the Maine Geological Survey). Together with the similarly-sized cladoxylopsid Pseudosporochnus, these were effectively the first trees - not much compared to their modern successors, perhaps, but very impressive compared to anything that came before them (with the exception, of course, of the primordial oddity Prototaxites). It is interesting to imagine what the environment of these early "forests" would have been like. How did they handle the weather, for a start? In the absence of a strong root system to anchor them down, were they prone to collapsing in the wind? If this was so, did they grow rapidly to compensate for their short lives, or did the rhizome readily send up new shoots to replace lost ones? (Remember, with no leaves either, the entire stem would have probably been photosynthetic.) How did this affect life for the early terrestrial animals taking advantage of their presence? There may have been the beginnings of a forest, but a world recognisably our own was still a long way off.
REFERENCES
Friedman, W. E., & M. E. Cook. 2000. The origin and early evolution of tracheids in vascular plants: integration of palaeobotanical and neobotanical data. Philosophical Transactions of the Royal Society of London Series B 355: 857-868.
Gerrienne, P. 1997. The fossil plants from the Lower Devonian of Marchin (northern margin of Dinant Synclinorium, Belgium): V. Psilophyton genseliae sp. nov., with hypotheses on the origin of Trimerophytina. Review of Palaeobotany and Palynology 98: 303-324.
Mauseth, J. D. 2008. Botany, 4th ed. Jones & Bartlett Publishers.
Selosse, M.-A., & F. Le Tacon. 1998. The land flora: a phototroph-fungus partnership? Trends in Ecology and Evolution 13 (1): 15-20.
Taylor, E. L., & M. Krings. 2008. Paleobotany, 2nd ed. Academic Press.

If you want to find out about the evolution of terrestrial life for vertebrates, there are countless sources out there for you to turn to. But if you want to find out about the evolution of terrestrial life for plants, then your options are probably much thinner. Which is just one more example of how screwed up our priorities as humans are, because there's no doubt which is the greater achievement. When the first terrestrial vertebrates emerged, they found a world already made lush by a covering of vegetation. But the first terrestrial plants would have found nothing waiting for them but bare, hostile rock*. It's amazing that they ever managed at all.
*To be honest, I lie slightly. In places where there was available moisture, I'm sure that a film of bacteria would have grown. Ditto for unicellular algae and other such organisms. If lichen-type associations were around at the time (and a cyanobacteria-zygomycete association is preserved in the Rhynie Chert - Selosse & Le Tacon, 1998), the world would have been their mollusc. Sadly, with little potential for their fossilisation and discovery, we may never really know about the contributions of these first unicellular pioneers.

But manage they did, and by the early Devonian the world was home to a small but respectable diversity of land plants. Most of the vascular plants of the time have been divided between the rhyniophytes, lycophytes, trimerophytes and cladoxylopsids (doubtless there were also moss- and liverwort-like plants around too, if not actual mosses and liverworts, but the spotty fossil record of bryophyte-grade plants doesn't quite reach that far back). Almost all of them, admittedly, would have been fairly similar to the non-expert eye - small, shrubby affairs with simple branching systems and no true leaves or roots. Examination of their fine structure (particularly of their vascular systems) is necessary to recognise their true affinities - rhyniophytes in the stem lineage for all vascular plants; lycophytes including the ancestors of modern Lycopodium, Selaginella and Isoetes; trimerophytes on the stem leading to modern ferns and seed plants; and cladoxylopsids on the stem of modern ferns*. Each of these groups quite possibly represents a grade rather than a clade, but in most cases it is not possible to actually demonstrate this one way or another.
*It is worth noting (especially in relation to a question asked in the comments to a previous post) that the vascular cells of rhyniophytes, lycophytes and trimerophytes each have distinct morphologies from each other** (Friedman & Cook, 2000), and this has led some authors to suggest that the vascular system may have developed independently in each of the three lineages. For now, though, it seems more parsimonious to assume a common origin followed by evolutionary divergence.
**It is also worth noting that when Friedman & Cook (2000) wrote their review, we actually knew more about the structure of the vascular cells in Devonian lycophytes and trimerophytes than in living lycophytes and ferns. Previous studies of vascular cell structure in living plants had almost exclusively looked at seed plants alone.
Trimerophytes differed from the more basal rhyniophytes in their mode of branching - whereas the basalmost land plants had branched dichotomously (dividing into two branches with each branch growing equivalently), trimerophytes branched anisotomously (one branch growing more than the other), effectively giving the trimerophytes some degree of a central stem (this process is called overtopping). Secondary branches from the central stem still branched dichotomously. Sporangia were borne on the tips of the branches, and at least some trimerophytes grew elongate sporangia in pairs that twisted around each other (Gerrienne, 1997).
 One particular trimerophyte, Psilophyton princeps, holds a particular significance for palaeobotany as the first Devonian plant to be reconstructed, by William Dawson in 1859 (Taylor & Krings, 2008), with a large creeping rhizome extending successive upright shoots. But perhaps even more significant was the size reached by some trimerophytes. While most Devonian vascular plants would have been struggling to reach half a metre in height, the trimerophyte Pertica dalhousii has been estimated to have reached up to three metres (Mauseth, 2008) - about the height of the ceiling of an average house (the related but smaller species Pertica quadrifaria is shown to the left, in a reconstruction from the Maine Geological Survey). Together with the similarly-sized cladoxylopsid Pseudosporochnus, these were effectively the first trees - not much compared to their modern successors, perhaps, but very impressive compared to anything that came before them (with the exception, of course, of the primordial oddity Prototaxites). It is interesting to imagine what the environment of these early "forests" would have been like. How did they handle the weather, for a start? In the absence of a strong root system to anchor them down, were they prone to collapsing in the wind? If this was so, did they grow rapidly to compensate for their short lives, or did the rhizome readily send up new shoots to replace lost ones? (Remember, with no leaves either, the entire stem would have probably been photosynthetic.) How did this affect life for the early terrestrial animals taking advantage of their presence? There may have been the beginnings of a forest, but a world recognisably our own was still a long way off.
One particular trimerophyte, Psilophyton princeps, holds a particular significance for palaeobotany as the first Devonian plant to be reconstructed, by William Dawson in 1859 (Taylor & Krings, 2008), with a large creeping rhizome extending successive upright shoots. But perhaps even more significant was the size reached by some trimerophytes. While most Devonian vascular plants would have been struggling to reach half a metre in height, the trimerophyte Pertica dalhousii has been estimated to have reached up to three metres (Mauseth, 2008) - about the height of the ceiling of an average house (the related but smaller species Pertica quadrifaria is shown to the left, in a reconstruction from the Maine Geological Survey). Together with the similarly-sized cladoxylopsid Pseudosporochnus, these were effectively the first trees - not much compared to their modern successors, perhaps, but very impressive compared to anything that came before them (with the exception, of course, of the primordial oddity Prototaxites). It is interesting to imagine what the environment of these early "forests" would have been like. How did they handle the weather, for a start? In the absence of a strong root system to anchor them down, were they prone to collapsing in the wind? If this was so, did they grow rapidly to compensate for their short lives, or did the rhizome readily send up new shoots to replace lost ones? (Remember, with no leaves either, the entire stem would have probably been photosynthetic.) How did this affect life for the early terrestrial animals taking advantage of their presence? There may have been the beginnings of a forest, but a world recognisably our own was still a long way off.REFERENCES
Friedman, W. E., & M. E. Cook. 2000. The origin and early evolution of tracheids in vascular plants: integration of palaeobotanical and neobotanical data. Philosophical Transactions of the Royal Society of London Series B 355: 857-868.
Gerrienne, P. 1997. The fossil plants from the Lower Devonian of Marchin (northern margin of Dinant Synclinorium, Belgium): V. Psilophyton genseliae sp. nov., with hypotheses on the origin of Trimerophytina. Review of Palaeobotany and Palynology 98: 303-324.
Mauseth, J. D. 2008. Botany, 4th ed. Jones & Bartlett Publishers.
Selosse, M.-A., & F. Le Tacon. 1998. The land flora: a phototroph-fungus partnership? Trends in Ecology and Evolution 13 (1): 15-20.
Taylor, E. L., & M. Krings. 2008. Paleobotany, 2nd ed. Academic Press.
Like, Wow. Just... Wow.
This is something I saw this morning at Small Things Considered, that I thought was just so spectacularly brilliant that I just had to copy it:

One hundred and sixty-seven species of ciliate, artfully arranged and all drawn to scale, from the gigantic Stentor to the sinuous Homalozoon to the infinitesimal Cinetochilum. The image comes from here, where not only can you see it in its full glory, but you'll find the key to the numbering that tells you what each one of these marvels is. The species are partially arranged in line with their chosen habitats - those towards the top left are found in the open water column in lakes, those around the centre of the bottom are anaerobes, while the others make their homes among sediment. And there's even little extra bits of detail hidden within - see if you can find the Chilodonella crawling along the Epistylis stalk, for instance. Enjoy!
Then if you're in the mood for more details on ciliates, including comments on their mind-blowingly complicated genetic system, take a look at my earlier posts here and here. And if anyone is feeling really generous and wants to get this printed out as a wall poster for me...

One hundred and sixty-seven species of ciliate, artfully arranged and all drawn to scale, from the gigantic Stentor to the sinuous Homalozoon to the infinitesimal Cinetochilum. The image comes from here, where not only can you see it in its full glory, but you'll find the key to the numbering that tells you what each one of these marvels is. The species are partially arranged in line with their chosen habitats - those towards the top left are found in the open water column in lakes, those around the centre of the bottom are anaerobes, while the others make their homes among sediment. And there's even little extra bits of detail hidden within - see if you can find the Chilodonella crawling along the Epistylis stalk, for instance. Enjoy!
Then if you're in the mood for more details on ciliates, including comments on their mind-blowingly complicated genetic system, take a look at my earlier posts here and here. And if anyone is feeling really generous and wants to get this printed out as a wall poster for me...
More Things in Heaven and Ocean
Following on from the post just put up on xenophyophores, I couldn't pass on without mentioning these fellows:

This is Haeckel's (1889) plate of members of the Ammoclathrinidae, taken from here. Haeckel described members of this family* among his "Deep-Sea Keratosa", the other examples of which have since been identified as xenophyophores (and not, as Haeckel thought they were, sponges). Like xenophyophores, ammoclathrinids had coverings constructed from foreign objects, and when I wrote the page on xenophyophores for Palaeos.com, I interpreted ammoclathrinids as being xenophyophores. True, Haeckel had originally identified separate cells inside his ammoclathrinids, but I suggested that this could have been an artefact caused by his use of acid to prepare the specimens, and they might have originally been coenocytic. However, when I wrote that I was working from Tendal's (1972) recounting of Haeckel's work, and hadn't seen the original description. Looking at it now, I have to admit that the illustrations in Haeckel's plate look very different from xenophyophores.
*Which he called Ammoconidae, but this name was changed by Tendal (1972) for reasons of preoccupation.
Unfortunately, Haeckel's original material no longer exists (it seems likely that he pulled it apart in the process of describing it), and no-one has ever laid eyes on a recognisable ammoclathrinid since. The nature of these organisms, therefore, is a complete mystery. Such a situation is more common than you may think, particularly in microbiology. The protistological literature is littered with examples of organisms seen and described by someone looking down a microscope in the 1800s, and never recorded since. It might be tempting to wonder whether the original describer ever really saw what they thought they saw at all, but that may be ignoring one of the major issues in microbiology itself - the world is very, very large, and the things being looked for a very, very small. If comparatively large mammal species can elude attention for numbers of years, how much more so can tiny micro-organisms? Or something like Ammoclathrinidae, lurking somewhere in the little-explored deep sea?
REFERENCES
Haeckel E. 1889. Report on the Deep-Sea Keratosa. Report on the Scientific Results of the Voyage of H. M. S. Challenger during the years 1873–76. Zoology 32 (part 82): 1–92.
Tendal, O. S. 1972. A monograph of the Xenophyophoria (Rhizopodea, Protozoa). Galathea Report 12: 7-99.

This is Haeckel's (1889) plate of members of the Ammoclathrinidae, taken from here. Haeckel described members of this family* among his "Deep-Sea Keratosa", the other examples of which have since been identified as xenophyophores (and not, as Haeckel thought they were, sponges). Like xenophyophores, ammoclathrinids had coverings constructed from foreign objects, and when I wrote the page on xenophyophores for Palaeos.com, I interpreted ammoclathrinids as being xenophyophores. True, Haeckel had originally identified separate cells inside his ammoclathrinids, but I suggested that this could have been an artefact caused by his use of acid to prepare the specimens, and they might have originally been coenocytic. However, when I wrote that I was working from Tendal's (1972) recounting of Haeckel's work, and hadn't seen the original description. Looking at it now, I have to admit that the illustrations in Haeckel's plate look very different from xenophyophores.
*Which he called Ammoconidae, but this name was changed by Tendal (1972) for reasons of preoccupation.
Unfortunately, Haeckel's original material no longer exists (it seems likely that he pulled it apart in the process of describing it), and no-one has ever laid eyes on a recognisable ammoclathrinid since. The nature of these organisms, therefore, is a complete mystery. Such a situation is more common than you may think, particularly in microbiology. The protistological literature is littered with examples of organisms seen and described by someone looking down a microscope in the 1800s, and never recorded since. It might be tempting to wonder whether the original describer ever really saw what they thought they saw at all, but that may be ignoring one of the major issues in microbiology itself - the world is very, very large, and the things being looked for a very, very small. If comparatively large mammal species can elude attention for numbers of years, how much more so can tiny micro-organisms? Or something like Ammoclathrinidae, lurking somewhere in the little-explored deep sea?
REFERENCES
Haeckel E. 1889. Report on the Deep-Sea Keratosa. Report on the Scientific Results of the Voyage of H. M. S. Challenger during the years 1873–76. Zoology 32 (part 82): 1–92.
Tendal, O. S. 1972. A monograph of the Xenophyophoria (Rhizopodea, Protozoa). Galathea Report 12: 7-99.
Living with Poo - A New Xenophyophore

And if you don't know what a xenophyophore is, then shame on you! Xenophyophores are sessile deep-sea protists that often reach comparatively gigantic sizes. One species, Stannophyllum venosum (which looks something like half a plate sitting on its edge), can be nearly a foot across (Tendal, 1972). Images from deep-sea submersibles have shown that xenos can be spectacularly abundant, carpeting the ocean floor in some places.
Xenophyophores are sometimes referred to as the largest unicellular organisms, but as I've mentioned before, that's arguably not entirely appropriate. Rather, xenophyophores have a coenocytic or hyphal organisation, with numerous nuclei scattered throughout long branching cytoplasmic tubes. The name "xenophyophore" means "bearer of foreign objects", and refers to the external test of the organism, which it constructs by cementing together objects it collects from the sediment around it - sand grains , for instance, or shells of other organisms - using a polysaccharide cement. The xenos also sequester their faecal pellets, which they may also integrate into their skeleton. Different species of xeno can be distinguished by the nature and arrangement of the foreign particles in the test (they are often quite picky about what they use), and the proportion of foreign particles to cement or faecal pellets. The species Cerelasma massa, for instance, differs from other xenos in using no foreign particles whatsoever, but only cement and its own faecal pellets - hence also being a contender for the title of most disgusting organism in existence. Species that use a high proportion of foreign particles in their construction are generally quite rigid, while those using more cement are softer.

Yesterday saw the publication of a new xenophyophore species, Shinkaiya lindsayi (the genus is named, offhand, after the submersible that was used to collect the type specimen) by Lecroq et al. (2009). One of the most significant features of the new paper is that it includes a molecular phylogenetic analysis of the new species. Xenophyophores have been sequenced on one occasion before, by Pawlowski et al. (2003), who placed the species Syringammina corbicula among basal Foraminifera. It is good to see that Lecroq et al. place Shinkaiya as the sister to Syringammina, and the two together fit in the same position among forams originally found by Pawlowski et al.. The closest relative to xenophyophores identified is a foram called Rhizammina, which is also sessile, constructs a test of foreign matter, and even sequesters faecal pellets in a similar manner to xenophyophores.
This is a noteworthy achievement - these are not easy organisms to sequence. Not only is there a shortage of accessible material, but xenos and forams both tend to have large numbers of bacteria and other micro-organisms living around them, just aching to contaminate DNA samples (the very first molecular phylogenetic analysis of a foram, for instance, suggested a close relationship between forams and dinoflagelates, only to have it later shown that the sequence analysed belonged not to the foram but to parasitic micro-organisms living in the foram*). So the fact that Lecroq et al.'s results are not only well-supported, but make a lot of intuitive sense morphologically, makes this a very nice study indeed.
*Still, if the recently suggested SAR clade is correct, there is a certain irony to this - forams may be somewhat related to dinoflagellates after all.

REFERENCES
Haeckel E. 1889. Report on the Deep-Sea Keratosa. Report on the Scientific Results of the Voyage of H. M. S. Challenger during the years 1873–76. Zoology 32 (part 82): 1–92.
Lecroq, B., A. J. Gooday, M. Tsuchiya & J. Pawlowski. 2009. A new genus of xenophyophores (Foraminifera) from Japan Trench: morphological description, molecular phylogeny and elemental analysis. Zoological Journal of the Linnean Society 156: 455-464.
Pawlowski, J., M. Holzmann, J. Fahrni & S. L. Richardson. 2003. Small subunit ribosomal DNA suggests that the xenophyophorean Syringammina corbicula is a foraminiferan. Journal of Eukaryotic Microbiology 50: 483-487.
Tendal, O. S. 1972. A monograph of the Xenophyophoria (Rhizopodea, Protozoa). Galathea Report 12: 7-99.
Southern Moss (Taxon of the Week: Ptychomitrium muelleri)

Mosses are often treated as the poor relation in plant diversity. Popular presentations of plant evolution often tend to have even more of a Scala Natura-esque slant to them than presentations of animal evolution, and so mosses and other non-vascular plants get glossed over as mere stepping stones to their more upright "descendants", if they even warrant a mention at all. This is, of course, complete rubbish - mosses have a very respectable diversity of species (about 10,000, according to Tree of Life). I've met a few moss researchers over the years, and a more devoted following a taxon could not hope for.
Ptychomitrium muelleri is a haplolepidous moss of the family Ptychomitriaceae (I'll explain what that means in a minute). It grows to a maximum height of one and a half centimetres, and if the type specimens are any indication, prefers to grow on rocks. Ptychomitrium mosses seem to be found more or less worldwide, but Ptychomitrium muelleri itself is found in south-eastern Australia, New Caledonia, southern South America and southernmost Africa. What is interesting about this species' distriution is that it was thought to be endemic to Australia until very recently when Cao et al. (2001) established that species described from each of the other localities were conspecific with P. muelleri. As a result, P. muelleri has what might be described as a classic "Gondwanan" distribution, but I rather doubt that Gondwana had anything to do with it. After all, moss spores are very light and extremely easily dispersed, and surely it is no coincidence that all the localities where P. muelleri can be found lie roughly along the same wind belt.

The diagram of a moss life cycle above has been stole from Palaeos.com. For the nonce, the important details are these - mosses, like other plants, are multicellular at both the haploid and diploid stages of the life cycle, but unlike vascular plants, the larger, dominant stage of the life cycle is the haploid gametophyte. When a female gametophyte is fertilised, the diploid sporophyte remains attached to the gametophyte and grows a spore-filled capsule that eventually breaks open (after the loss of the protective calyptra) to release the spores. Around the mouth of the capsule is a ring of "teeth", the peristome. In basal mosses, the peristome is made up of entire cells, but in the class Bryopsida, the arthrodontous mosses (which includes the larger part of the mosses), the teeth are reduced to cell wall remnants. Most of the bryopsid lineages have two rows of teeth in the peristome, an outer and an inner, but Ptychomitrium belongs to a group called the Haplolepideae or Dicranidae which have only the inner row of teeth. The haplolepidous mosses form a monophyletic clade within the Bryopsida.

Most of the features separating moss taxa are microscopic and relate to such things as cell arrangement (which is my weaselly method of saying that I don't understand a word of them), but Ptychomitrium is distinguished by having a mitrate calyptra (and those unsure what "mitrate" means might want to look at the Taxon of the Week post of two weeks ago) with characteristic lobes around the lower edge (Hernández-Maqueda et al., 2008, compare it to a Hawaiian skirt). Ptychomitrium muelleri has (amongst other features) lingulate (tongue-shaped, I'm guessing) leaves with smooth margins, and ovoid capsules.
REFERENCES
Cao, T., S. Guo & Y. Zhang. 2001. Distribution of Ptychomitrium muelleri (Bryopsida), with its synonyms. The Bryologist 104 (4): 522-526.
Hernández-Maqueda, R., D. Quandt, O. Werner & J. Muñoz. 2008. Phylogeny and classification of the Grimmiaceae/Ptychomitriaceae complex (Bryophyta) inferred from cpDNA. Molecular Phylogenetics and Evolution 46 (3): 863-877.
Knocked Off the Perch (Taxon of the Week: Percidae)

Okay, this post has been delayed again. It's been an unusual week, is all I can say. I'd tell you all about, but I have very good reasons to believe that that would be extremely dull.
In earlier posts, I have ranted in a rather esoteric manner about my distaste with the commonly recognised fish order "Perciformes", really a random multi-paraphyletic assemblage of the more generalised members of the clade Percomorpha. In the recent partial reclassification of the Percomorpha by Li et al. (2009), the name "Perciformes" was ditched entirely, and the clade containing the family Percidae was instead called Serraniformes (suggesting, offhand, that some sort of taxonomic karma is dooming this taxon to be associated with confusing names - the family Serranidae as commonly recognised itself seems likely to be polyphyletic, and a number of "serranids" are not guaranteed Serraniformes). But even before the Perciformes of common use were recognised as a wastebasket assemblage (if, indeed, there ever really was such a time), Percidae was always a slightly odd choice for the type family. The Percidae, the perches and darters, are not particularly average Perciformes.
Among the percomorphs, percids are unusual for one main reason - they're almost entirely freshwater (a few European species stray into brackish waters, but only one species - Sander marinus, the estuarine perch of the Black and Caspian Seas - is a permanent resident in them). While the percomorphs have achieved true world dominance in the upper parts of the ocean, including the vast majority of coastal and surface-pelagic fish species, they have never made such significant inroads into fresh water. A few percomorph lineages have been very successful in fresh water, such as the Cichlidae, the Anabantiformes and various members of the Smegmamorpha*. But in contrast to their surface-marine monopoly, percomorphs have to share dominance of the fresh-water environment with members of the clade Otophysi - Cypriniformes, Characiformes and Siluriformes.
*No, honestly, it's a real name.

The Serraniformes also include the Trachinidae (weevers), the circum-Antarctic notothenioids and the majority of what were the Scorpaeniformes. Relationships within the Serraniformes are yet to be hammered out, but the Percidae probably divide from the others reasonably basally. Ten genera of living Percidae are currently recognised, with more than two hundred species. Phylogenetic analysis of the family by Sloss et al. (2004) recognised three main clades of unresolved relationships - the Holarctic genus Perca, the mostly Eurasian clade of Gymnocephalus plus Luciopercinae (genera Romanichthys, Sander and Zingel, with three species of Sander in North America), and the North American clade of Etheostomatinae (Ammocrypta, Crystallaria, Etheostoma and Percina). [The tenth genus includes the single uncommon species Percarina demidoffi of rivers running into the Black Sea, and was not analysed by Sloss et al. due to lack of material. Percarina was previously classified in the possibly non-monophyletic Percinae* with Perca and Gymnocephalus and differs from most other Percinae in spawning in brackish waters, so establishing its relationships would be very interesting.] While the greater phylogenetic disparity of Percidae is concentrated in the western Palaearctic and the family is believed to have originated in that area, the greater diversity of species is definitely found in North America. Well over two-thirds of percid species belong to the Etheostomatinae, with the greater part of those in the genus Etheostoma (which, however, may not be monophyletic).
*Though the non-monophyly of Percinae found by Sloss et al. is in contrast to their breeding behaviour - Percinae differ from other percids in laying their eggs encased in long gelatinous strands, while Luciopercinae and (ancestrally) Etheostomatinae are broadcast spawners.

Human interest in the Percidae (as with most matters, really) has usually been related to one of two things - eating or sex. The larger percids of the "Percinae" and Luciopercinae are widely caught for food, and the European perch Perca fluviatilis has been introduced to many localities outside its native range such as New Zealand for the amusement of anglers. Some percids, such as the walleye Sander vitreus, have been recorded reaching lengths of over a metre (though such sizes are, of course, exceptional - a more average walleye would be about twenty centimetres). Species of the Etheostomatinae, known as darters, are not targets of fishing - members of this subfamily (as well as some species of Luciopercinae) are smaller than other percids, less than ten centimetres in length*, and wouldn't offer much in the way of eating. Still, darters more than make up their interest in the other regard of sex. They show a wide diversity of breeding behaviour, from broadcast spawners to some that bury their eggs in sediment or gravel to species that lay their eggs safely hidden on the underside of rocks. Other species may glue their eggs to vegetation (Winn, 1958a, b). During the breeding season, most (but not all) darters move from deeper to shallower waters (many species favour riffle areas) where the males usually establish a breeding territory (as reported by Winn, 1958b, the presence of other males seems to be required to incite the successful establishment of a territory - solitary males tended to lose interest in a potential territory and wander off). Some darter species are fairly relaxed about their territories and only fend off males of their own species, but other darters may be decidedly pugnacious and attack just about anything that moves. Challenging males approach each other with fins held high, and their colours will often become brighter. They may circle each other and butt or bite at each other's tail regions. After a male has mated with a female and she has laid her eggs, he may or may not remain in the area to guard them. Experiments have shown that if the eggs are removed or replaced, the male continues to guard the same spot, so it is the territory that induces guarding behaviour rather than the presence of eggs. Hybrids have been recorded between a number of darter species and seem to be not uncommon, especially where species have been spread outside their native range (Stauffer et al., 1995).
*As a corollary of their smaller size, it is worth noting that darters (and the smaller Luciopercinae) also lack swim bladders.

REFERENCES
Reeves, C. D. 1907. The breeding habits of the rainbow darter (Etheostoma cœruleum Storer), a study in sexual selection. Biological Bulletin 14 (1): 35-59.
Sloss, B. L., N. Billington & B. M. Burr. 2004. A molecular phylogeny of the Percidae (Teleostei, Perciformes) based on mitochondrial DNA sequence. Molecular Phylogenetics and Evolution 32 (2): 545-562.
Stauffer, J. R., Jr, J. M. Boltz & L. R. White. 1995. The fishes of West Virginia. Proceedings of the Academy of Natural Sciences of Philadelphia 146: 1-389.
Winn, H. E. 1958a. Observation on the reproductive babits of darters (Pisces-Percidae). American Midland Naturalist 59 (1): 190-212.
Winn, H. E. 1958b. Comparative reproductive behavior and ecology of fourteen species of darters (Pisces-
Percidae). Ecological Monographs 28 (2): 155-191.
Subscribe to:
Posts (Atom)















