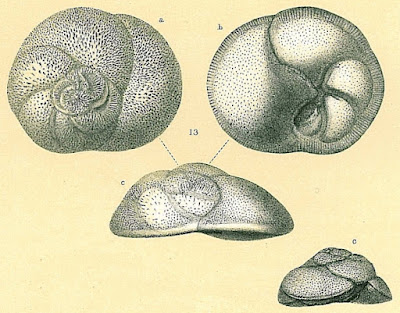
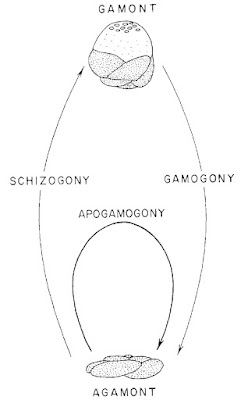
Rosalinids may be regarded as fairly typical-looking marine rotaliids with the test growing freely as a low trochospire (so a flattened cone or dish shape). The aperture of the test is a low slit on the interior margin along the umbilicus (Hansen & Revets 1992). Rosalinids have a complex life cycle involving both benthic and planktonic stages (Sliter 1965). The asexually reproducing diploid stage is benthic. Depending on conditions, diploid individuals may divide to produce other diploid individuals, resulting in several asexual generations. Eventually, however, the diploid generation will undergo meiosis to produce the haploid sexual generation (in the common species Rosalina globularis, this is induced by exposure to warmer water). In the sexual generation, a large globular chamber forms at maturity that covers the umbilical side of the test. This float chamber becomes filled with gas, allowing the foram to disperse planktonically before releasing gametes to produce the next diploid generation. Planktonic individuals are distinct enough in appearance from their benthic counterparts that they were long mistaken for distinct taxa before their identity was revealed by lab cultures.
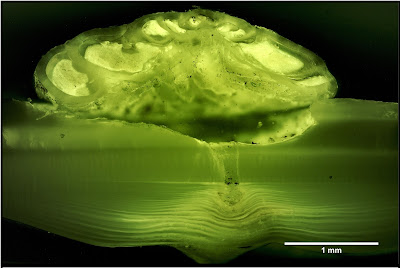
The majority of forams are particulate feeders. A network of filamentous pseudopodia radiating outwards from the cell body captures micro-organisms and other organic particles. However, one genus of rosalinids, Hyrrokkin, lives as parasites on sessile invertebrates (Cedhagen 1994). Species of this genus have variously been found on sponges, corals and bivalves. On sponges, they settle on the inhalent surface of the sponge and dissolve the underlying tissues. On bivalves, they form pits on the shell surface from which they bore holes through to the body cavity. Pseudopodia extended through this hole allow the foram to feed on host tissue. Infested hosts may bear multiple scars from the foram moving about on the outer surface. The forams may also feed on other animals such as polychaete worms or bryozoans attached to the surface of their primary host. In such cases, Hyrrokkin remains in its original pit but develops an irregularly shaped chamber with its aperture directed towards the alternate prey. Hyrrokkin species evidently do well from their rapacious lifestyle: whereas other rosalinids are only a fraction of a millimetre in diameter, Hyrrokkin sarcophaga is an absolute giant reaching around six millimetres across and with protoplasm containing thousands of nuclei. Proving once again that one may make a great deal of profit from the labour of others.
REFERENCES
Cedhagen, T. 1994. Taxonomy and biology of Hyrrokkin sarcophaga gen. et sp. n., a parasitic foraminiferan (Rosalinidae). Sarsia 79: 65–82.
Hansen, H. J., & S. A. Revets. 1992. A revision and reclassification of the Discorbidae, Rosalinidae, and Rotaliidae. Journal of Foraminiferal Research 22 (2): 166–180.
Sliter, W. V. 1965. Laboratory experiments on the life cycle and ecologic controls of Rosalina globularis d'Orbigny. Journal of Protozoology 12 (2): 210–215.